Eurofins U.S. Clinical Diagnostics Network Receives EUA Approval of At Home COVID-19 Test
19 Octobre 2020 - 7:05AM
Business Wire
At Home COVID-19 Nasal PCR Test is easy to use
and painless
Eurofins U.S. Clinical Diagnostics, announced that Clinical
Enterprise Inc. has received an Emergency Use Authorization (EUA)
from the U.S. Food and Drug Administration (FDA) for their at Home
COVID-19 Nasal PCR Test. The EUA authorized self-collection kit
gives consumers a minimally invasive, convenient and quick option
to test from the comfort of their home. Results are provided via
email within 24 hours of sample receipt.
Available through the Eurofins direct-to-consumer company,
empowerDX, the at home test can be ordered online
(https://empowerdxlab.com/) for $89. After completing a
questionnaire, consumers will receive the sample collection kit via
FedEx.
Eurofins’ infectious disease centre for excellence, Viracor,
developed the test based on its highly sensitive, FDA
EUA-authorized SARS-CoV-2 RT-PCR assay.
The Eurofins U.S. Clinical Diagnostics network has been at the
forefront of COVID-19 testing – launching its first RT-PCR assay
for SARS-CoV-2 on March 13. Since then, Eurofins has rolled out
several highly sensitive and specific assays to thousands of
clients nationwide, including hospitals, physicians’ offices,
nursing homes, government entities, sports organizations and
employers. At-home COVID-19 testing adds additional capability to
an already robust menu including pooled testing, antibody testing,
wastewater, used-mask, and soon to be launched saliva testing.
Concurrent to ongoing COVID-19 assay innovation, Eurofins has
increased capacity throughout its network of labs to ensure
sufficient capacity is available for consistency of turnaround time
should COVID-19 infections continue to increase in the upcoming
weeks. Since March, Eurofins US Clinical Diagnostics testing
capacity has ramped up to over 500,000 samples per week. The
turnaround time for results has averaged less than18 hours from
receipt in the lab.
The at Home COVID-19 Nasal PCR Test is currently not available
in New York, New Jersey, Rhode Island, and Maryland.
This home-collection kit has not been FDA cleared or approved;
rather it has been authorized by FDA under an EUA only for the
self-collection and maintenance of nasal swab specimens as an aid
in detection of nucleic acid from SARS-CoV-2, and not for any other
viruses or pathogens, and only for the duration of the declaration
that circumstances exist justifying the authorization of emergency
use of in medical devices during the COVID-19 outbreak under
Section 64(b)(1) of the Federal Food, Drug and Cosmetic Act, 21
U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or
revoked sooner.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201018005083/en/
Matt Urbanek ir@eurofins.com
Eurofins Scientific (EU:ERF)
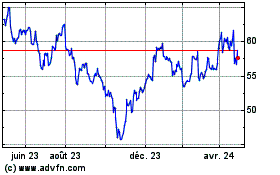
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
Eurofins Scientific (EU:ERF)
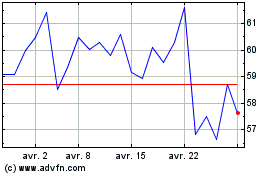
Graphique Historique de l'Action
De Avr 2023 à Avr 2024