Medidata Announces Launch of Medidata Detect, its Centralized Statistical Monitoring Solution for Clinical Trials
13 Août 2020 - 4:01PM
Business Wire
Medidata, a Dassault Systèmes company, announced the launch of
Medidata Detect, a centralized statistical monitoring solution that
improves data quality and promotes patient safety in clinical
trials for new medicines, vaccines and medical devices. Medidata
Detect, part of Medidata’s industry-leading, regulatory-compliant
and unified Medidata Rave Clinical Cloud platform, helps customers
manage data quality, monitor site performance and promote patient
safety by uncovering and finding errors, trends and anomalies in
data through statistical algorithms and tests.
“Medidata Detect is a new and vital machine learning tool
designed to improve data integrity and reduce trial risk,” said
Glen de Vries, co-founder and co-CEO, Medidata. “Detect is another
example of our continuous innovation that helps advance clinical
trials with increased data accuracy and speed. It’s another way
we’re working to get important new vaccines and therapeutics to
patients in safer, faster ways.”
A report in the Journal of the American Medical Society (JAMA)*
found almost a quarter of new drug submissions required one or more
resubmissions before they were approved, with a median delay to
approval of 435 days following the first unsuccessful submission.
Medidata Detect uses one central system for aggregation and review
of any number of data sources, flagging data errors, trends, and
anomalies in real time. This translates into better quality data
and decisions throughout the trial, and shortens each cycle review
time by weeks.
Medidata Detect identifies known and unknown risks, and triggers
corrective actions, which proactively minimize study delays and
submission failures in trials. A foundational step in adopting a
RBQM (risk-based quality management) approach to clinical
development, Medidata Detect is able to define and manage study
risks, expose unanticipated data anomalies and cut down on
programmers’ time and effort through proprietary machine learning
algorithms that require no customer programming.
Medidata is a wholly owned subsidiary of Dassault Systèmes,
which with its 3DEXPERIENCE platform is positioned to lead the
digital transformation of life sciences in the age of personalized
medicine with the first end-to-end scientific and business
platform, from research to commercialization.
* Sacks LV, Shamsuddin HH, Yasinskaya YI, Bouri K, Lanthier ML,
Sherman RE, “Scientific and Regulatory Reasons for Delay and Denial
of FDA Approval of Initial Applications for New Drugs, 2000-2012.”
JAMA.2014;311(4):378–384.
About Medidata
Medidata is leading the digital transformation of life sciences,
creating hope for millions of patients. Medidata helps generate the
evidence and insights to help pharmaceutical, biotech, medical
device and diagnostics companies, and academic researchers
accelerate value, minimize risk, and optimize outcomes. More than
one million registered users across 1,500 customers and partners
access the world's most-used platform for clinical development,
commercial, and real-world data. Medidata, a Dassault Systèmes
company (Euronext Paris: #13065, DSY.PA), is headquartered in New
York City and has offices around the world to meet the needs of its
customers. Discover more at www.medidata.com and follow us
@Medidata, The Operating System for Life Sciences™.
Medidata and Medidata Rave are registered trademarks of Medidata
Solutions, Inc., a wholly owned subsidiary of Dassault
Systèmes.
About Dassault Systèmes
Dassault Systèmes, the 3DEXPERIENCE Company, is a catalyst for
human progress. We provide business and people with collaborative
3D virtual environments to imagine sustainable innovations. By
creating virtual experience twins of the real world with our
3DEXPERIENCE platform and applications, our customers push the
boundaries of innovation, learning and production. Dassault
Systèmes brings value to more than 270,000 customers of all sizes,
in all industries, in more than 140 countries. For more
information, visit www.3ds.com.
3DEXPERIENCE, the Compass icon, the 3DS logo, CATIA, BIOVIA,
GEOVIA, SOLIDWORKS, 3DVIA, ENOVIA, EXALEAD, NETVIBES, MEDIDATA,
CENTRIC PLM, 3DEXCITE, SIMULIA, DELMIA, and IFWE are commercial
trademarks or registered trademarks of Dassault Systèmes, a French
“société européenne” (Versailles Commercial Register # B 322 306
440), or its subsidiaries in the United States and/or other
countries.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200813005535/en/
Caroline Drucker Senior Director, Head of Corporate
Communications +1-347-675-3222 cdrucker@medidata.com
Paul Oestreicher Head of External Communications, Senior Writer
+1-917-536-3523 poestreicher@medidata.com
Dassault Systemes (EU:DSY)
Graphique Historique de l'Action
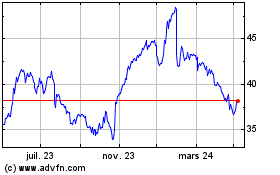
De Mar 2024 à Avr 2024
Dassault Systemes (EU:DSY)
Graphique Historique de l'Action
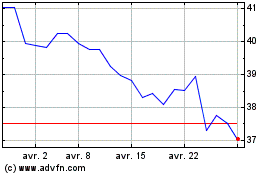
De Avr 2023 à Avr 2024