Neovacs to host KOL Meeting to discuss its therapeutic vaccin IFNalpha Kinoid for lupus treatment - February 11th Paris
05 Février 2019 - 7:30AM

Neovacs to host KOL Meeting to discuss its therapeutic vaccin
IFNalpha Kinoid for lupus treatment - February 11th Paris
NEOVACS TO HOST KOL MEETING TO
DISCUSS
ITS THERAPEUTIC VACCINE
IFNα KINOID FOR LUPUS
TREATMENT
FEBRUARY 11, 2019
– 17h00Hôtel Renaissance Le Parc Trocadéro – Paris
Paris and Boston, February 5th 2019 –
7h30 CET - Neovacs (Euronext Growth Paris: ALNEV), leader
in active immunotherapy for the treatment of auto-immune and
inflammatory diseases announces an event with Key Opinion Leaders
specializing in auto-immune diseases and rheumatology to discuss
its ground breaking product, the IFNα Kinoid for lupus
treatment.
- Professor Frederic Houssiau - Vice-Rector of
the Health Sciences Sector, at the University Catholic of Louvain,
Brussels, Belgium, formerly Head of the Rheumatology Department at
the Cliniques Universitaires Saint-Luc in Brussels. He is a
founding member of the Lupus Nephritis Trials Network and is
currently coordinating the trial with IFN-K.
- Professor Eric Morand - Head of the School of
Clinical Sciences at Monash University in Australia, He
is a clinical rheumatologist, and Head of the Monash Health
Rheumatology Unit.
The experts will present the natural history,
the symptoms and current prevalence of lupus. There is currently no
satisfactory treatment for this highly complex auto-immune
disease.
Professor Houssiau will present
the topline results of the Phase IIb obtained with IFNα Kinoid in
lupus (with which an immune response was achieved with 91% of
patients treated as well as post hoc results). They will discuss
the complex nature of the clinical scores used for lupus as well as
the clinical perspectives for doctors and their patients to use the
candidate therapeutic vaccin IFNα Kinoid.
Neovacs’ management will present the next
clinical steps and regulatory process required to enter into Phase
III.
Contacts
|
NeovacsCorporate
Communication& Investor Relations
Charlène Masson+33 1 53 10
93 00cmasson@neovacs.com |
Relations Presse – NewCapAnnie-Florence
LoyerTél : +33 (0)6 88 20 35 59 / +33 (0)1 44 71 00
12afloyer@newcap.fr Léa JacquinTel :
+33(0)1 44 71 20 41ljacquin@newcap.fr |
ORPHEON FINANCEFinancial Communication and
Investor Relations James
Palmer+33 7 60 92 77 74j.palmer@orpheonfinance.com
|
About IFNα
Kinoid
Neovacs anti-IFNα therapy consists of patient’
active immunization using Interferon α (IFNα) kinoid (IFNα Kinoid).
IFNα Kinoid is a heterocomplex consisting of an inactivated IFNα
coupled to a T-helper stimulating carrier protein, Keyhole Limpet
Hemocyanin (KLH). IFN-K is emulsified with Montanide™ oily adjuvant
that non-specifically stimulates cell-mediated immune (CMI)
responses to antigens. IFNα Kinoid elicits the production of
neutralizing polyclonal antibodies directed against the excess
IFNα, thus blocking its ability to activate the inflammatory
cascade. The generation of polyclonal neutralizing antibodies
against IFNα following the administration of IFNα Kinoid is
relevant to diseases mediated by IFNα over-production, such as
Systemic Lupus Erythematosus (SLE), Dermatomyositis (DM), Type I
Diabetes (T1D) and Sjögren’s Syndrome (SS).
About Lupus Systemic lupus
erythematosus (SLE) or lupus erythematosus is a debilitating,
chronic autoimmune disease whose etiology remains unknown. SLE is
characterized by a loss of tolerance of self-antigens, with the
production of autoantibodies, especially antinuclear antibodies
that attack healthy tissues and cause inflammatory reactions in
different parts of the body. The disease can affect multiple organs
(skin, kidneys, joints, heart, lungs, central nervous system, etc.)
and is characterized by heterogeneous clinical signs (skin rashes,
arthritis, photosensitivity, nephritis, neurological disorders,
anemia, thrombocytopenia, etc.), which vary from one person to
another and change during the progression of the disease. Systemic
lupus erythematosus affects mostly women.
About NeovacsListed on Euronext
Growth since 2010, Neovacs is today a leading biotechnology company
focused on an active immunotherapy technology platform (Kinoids)
with applications in autoimmune and/or inflammatory diseases. On
the basis of the company’s proprietary technology for inducing a
polyclonal immune response (covered by four patent families that
potentially run until 2032) Neovacs is focusing its clinical
development efforts on IFNα-Kinoid, an immunotherapy being
developed for the indication of lupus, dermatomyositis and also in
preclinical trial for Type 1 diabetes. Neovacs is also conducting
preclinical development works on other therapeutic vaccines in the
fields of auto-immune diseases, oncology and allergies. The goal of
the Kinoid approach is to enable patients to have access to safe
treatments with efficacy that is sustained in these life-long
diseases. www.neovacs.fr
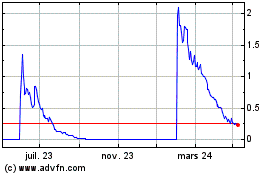
Neovacs (EU:ALNEV)
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
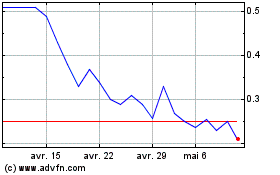
Neovacs (EU:ALNEV)
Graphique Historique de l'Action
De Avr 2023 à Avr 2024