Sanofi: FDA Approves Fluzone High-Dose Quadrivalent for Adults 65 and Older
05 Novembre 2019 - 12:14AM
Dow Jones News
By Stephen Nakrosis
Sanofi on Monday said the U.S. Food and Drug Administration
approved a supplemental Biologics License Application for its
Fluzone influenza vaccine for use by adults 65 and older.
The application was for the company's Fluzone High-Dose
Quadrivalent (Influenza Vaccine), Sanofi said.
"Fluzone High-Dose Quadrivalent is given to people 65 years of
age and older to help prevent influenza disease caused by influenza
A and B strains contained in the vaccine," the company said.
David Loew, Sanofi Executive Vice President and head of Sanofi
Pasteur, said, "We are excited to build upon the success of
trivalent Fluzone High-Dose with this FDA approval to expand
protection for an additional B strain. We have submitted filings
with additional regulatory bodies outside the U.S. and anticipate
approval in the European Union next spring."
--Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
November 04, 2019 17:59 ET (22:59 GMT)
Copyright (c) 2019 Dow Jones & Company, Inc.
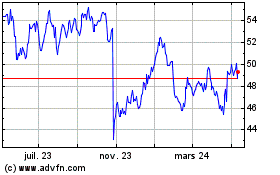
Sanofi (NASDAQ:SNY)
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
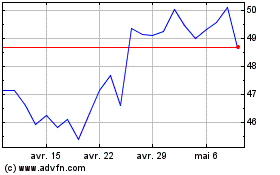
Sanofi (NASDAQ:SNY)
Graphique Historique de l'Action
De Avr 2023 à Avr 2024