AIM ImmunoTech Secures New State-of-the-Art Facility for Product Development and Testing
21 Juin 2022 - 2:35PM

AIM ImmunoTech Inc. (NYSE: American
AIM) (“AIM” or the “Company”), an immuno-pharma
company focused on the research and development of therapeutics to
treat multiple types of cancers, immune disorders, and viral
diseases, including COVID-19, the disease caused by the SARS-CoV-2
virus, announced today it has entered into a lease agreement with
the New Jersey Economic Development Authority for a 5,210 square
foot R&D facility at the New Jersey Bioscience Center (“NJBC”).
AIM Chief Executive Officer Thomas K. Equels
commented, “The AIM team is pleased to join a number of thriving,
prestigious life sciences companies at the NJBC. This
state-of-the-art facility will help us to continue to advance our
research and development of Ampligen to treat multiple types of
cancers, immune disorders, and viral diseases. We have made a
significant investment in new equipment for this facility and
believe that this space will enhance our future development
progress.”
The New Jersey Bioscience Center is a 50-acre
research park home to a number of successful biotech businesses.
The Incubator at North Brunswick is the leading incubator for small
businesses in the region dedicated to life sciences and
biotechnology companies. The NJBC has invested over $70 million in
facilities and improvements to its campus.About AIM
ImmunoTech Inc.
AIM ImmunoTech Inc. is an immune-pharma company
focused on the research and development of therapeutics to treat
multiple types of cancers, immune disorders, and viral diseases,
including COVID-19. The Company’s lead product, Ampligen®
(rintatolimod) is an immuno-modulator with broad spectrum activity
being developed for globally important cancers, viral diseases and
disorders of the immune system.
Ampligen is currently being used as a
monotherapy to treat pancreatic cancer patients in an Early Access
Program (EAP) approved by the Inspectorate of Healthcare in the
Netherlands at Erasmus Medical Center and AIM plans to initiate a
Phase 2 clinical study in 2022. The Company also has multiple
ongoing clinical trials to evaluate Ampligen as a combinational
therapy for the treatment of a variety of solid tumor types both
underway and planned at major cancer research centers.
Additionally, Ampligen is approved in Argentina for the treatment
of Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and
is currently being evaluated in many aspects of SARS-CoV-2/COVID-19
treatments and COVID-19 Long Hauler treatment.
For more information, please
visit aimimmuno.com and connect with the Company
on Twitter, LinkedIn, and Facebook.
Cautionary Statement
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995 (the “PSLRA”). Words such as “may,” “will,”
“expect,” “plan,” “anticipate” and similar expressions (as well as
other words or expressions referencing future events or
circumstances) are intended to identify forward-looking statements.
Many of these forward-looking statements involve a number of risks
and uncertainties. Among other things, for those statements, the
Company claims the protection of safe harbor for forward-looking
statements contained in the PSLRA. The Company does not undertake
to update any of these forward-looking statements to reflect events
or circumstances that occur after the date hereof.
Investor Relations ContactJTC
Team, LLCJenene Thomas 833-475-8247AIM@jtcir.com
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/6920d9fe-a3f8-4590-9d35-895e1be8af0d
AIM ImmunoTech (AMEX:AIM)
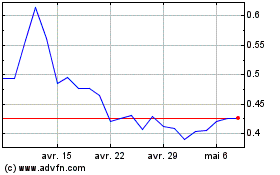
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
AIM ImmunoTech (AMEX:AIM)
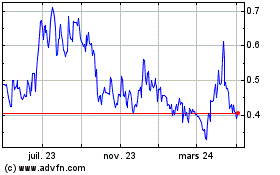
Graphique Historique de l'Action
De Avr 2023 à Avr 2024