AIM ImmunoTech Announces Abstract Accepted for Poster Presentation at the British Society for Immunology Congress 2022
02 Décembre 2022 - 4:45PM

AIM ImmunoTech Inc. (NYSE American: AIM)
(“AIM” or the “Company”), an immuno-pharma company focused on the
research and development of therapeutics to treat multiple types of
cancers, immune disorders, and viral diseases, including COVID-19,
the disease caused by the SARS-CoV-2 virus, today announced its
abstract has been accepted for poster presentation at the British
Society for Immunology Congress 2022 being held December 5-8, 2022
in Liverpool, UK.
Details for the presentation are as follows:
Poster No: P-220Title: Safety,
tolerability and biological activity of repeated intranasal
administration of TLR3 agonist Ampligen (Poly I:Poly C12U) in
healthy subjectsPresenter: Lisanne C.A. Smidt -
Centre for Human Drug Research, Leiden, the
NetherlandsSession: Poster Session 2Date
& Time: Wednesday December 7, 2022 from 5:30-7:00 PM
GMT
For more information, please visit the Congress
website, here.
About AIM ImmunoTech Inc.
AIM ImmunoTech Inc. is an immuno-pharma company
focused on the research and development of therapeutics to treat
multiple types of cancers, immune disorders, and viral diseases,
including COVID-19. The Company’s lead product, Ampligen®
(rintatolimod) is an immuno-modulator with broad spectrum activity
being developed for globally important cancers, viral diseases and
disorders of the immune system.
Ampligen is currently being used as a
monotherapy to treat pancreatic cancer patients in an Early Access
Program (EAP) approved by the Inspectorate of Healthcare in the
Netherlands at Erasmus Medical Center and AIM has commenced a Phase
2 clinical study in 2022. The Company also has multiple ongoing
clinical trials to evaluate Ampligen as a combinational therapy for
the treatment of a variety of solid tumor types both underway and
planned at major cancer research centers. Additionally, Ampligen is
approved in Argentina for the treatment of severe chronic fatigue
syndrome (CFS) and is currently being evaluated in many aspects of
SARS-CoV-2/COVID-19 myalgic encephalomyelitis/chronic fatigue
syndrome (ME/CFS) and Post COVID Conditions.For more information,
please visit aimimmuno.com and connect with the Company on Twitter,
LinkedIn, and Facebook.
Cautionary Statement
This press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995 (the “PSLRA”). Words such as “may,” “will,”
“expect,” “plan,” “anticipate” and similar expressions (as well as
other words or expressions referencing future events or
circumstances) are intended to identify forward-looking statements.
Many of these forward-looking statements involve a number of risks
and uncertainties. Among other things, for those statements, the
Company claims the protection of safe harbor for forward-looking
statements contained in the PSLRA. The Company does not undertake
to update any of these forward-looking statements to reflect events
or circumstances that occur after the date hereof.
Investor Relations Contact
JTC Team, LLC
Jenene Thomas
833-475-8247
AIM@jtcir.com
AIM ImmunoTech (AMEX:AIM)
Graphique Historique de l'Action
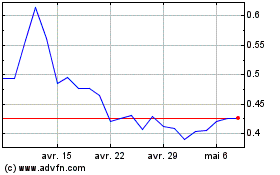
De Mar 2024 à Avr 2024
AIM ImmunoTech (AMEX:AIM)
Graphique Historique de l'Action
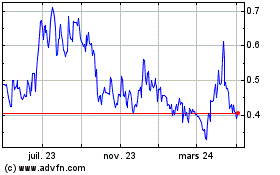
De Avr 2023 à Avr 2024