Theraclion Announces First Patient Inclusion in Early-Stage Breast Cancer Study
08 Mars 2022 - 9:12PM
Business Wire
Regulatory News:
THERACLION (ISIN: FR0010120402; Mnemo: ALTHE), an innovative
company developing a scalable robotic platform for non-invasive
echotherapy, announces today the first patient treatment in an
early-stage breast cancer study in collaboration with the
University of Virginia Cancer Center, USA. The trial seeks to
investigate the combination of High Intensity Focused Ultrasound
(HIFU) and low-dose chemotherapy to augment immune control of
breast cancer.
Early-stage breast cancer focus following promising results
of initial study
Registered on ClinicalTrials.gov, the new study was designed by
Pr. David Brenin and Pr. Patrick Dillon. The research aims to
evaluate the conjunctive effect of Theraclion’s focused ultrasound
and low-dose gemcitabine in early-stage breast cancer patients. ’We
will examine whether the dual therapy decreases myeloid-derived
suppressor cells and thus increases the immune response,’ explains
David Brenin, Chief of the Division of Breast and Melanoma Surgical
Services and Professor in the Department of Surgery at UVA Health.
‘If demonstrated to be effective, the combined treatment could
provide patients with early-stage breast cancer an improved chance
of survival,’ adds Patrick Dillon, Associate Professor in the
Department of Hematology and Oncology at UVA Health.
After an initial successful collaboration on late-stage breast
cancer patients, the University of Virginia (UVA) Cancer Center has
extended the use of Theraclion’s world-class technology to a
phase-2 investigation in early-stage breast cancer. These two
studies emphasize the need for a mixed approach, combining the
non-invasive HIFU, data-driven platform with the pharmacological
options, either chemotherapy or immune-oncology molecules, to offer
alternative treatments for early- and late-stage breast cancer.
Every year, 2.3 million women are diagnosed with breast cancer
and the disease is responsible for 685,000 deaths globally.
Moreover, 7.8 million women alive had been diagnosed with breast
cancer over the past 5 years as of 2021, making it the world’s most
prevalent cancer.1 An estimated 3.8 million women have a history of
breast cancer in the US2. ‘We are excited about this opportunity to
explore, together with the University of Virginia, our technology’s
full potential for improving patients’ lives’, says David
Caumartin.
Theraclion’s strategic development in 3 major therapeutic
areas in 3 geographies
Theraclion’s growth strategy involves expanding 3 major
therapeutic areas in 3 geographic areas:
- Varicose veins, in the USA (seeking FDA clearance) and in
Europe
- Thyroid, in Europe and in China
- Breast cancer, mostly in the USA
To achieve these ambitious goals, Theraclion will secure
financing and strategic partnerships and mainly focus on key
technical and clinical milestones.
About Theraclion
At Theraclion we believe that surgery, as we know it, is
outdated. It converts optimistic patients into anxious individuals,
brilliant doctors into exhausted system executors and stretches
healthcare systems to the limit. We have disrupted this convention
by creating extracorporeal treatment platforms. We replace surgery
with a robotic treatment from outside the body using High Intensity
Focused Ultrasound (HIFU). Our leading edge echotherapy platforms
are currently CE marked in non-invasive treatment of varicose veins
with SONOVEIN® and of breast fibroadenomas and thyroid nodules with
ECHOPULSE®.
Located in Malakoff, near Paris, our employees live and breathe
innovation by extensive clinical research and harness artificial
intelligence. The market of varicose veins treatment alone requires
around 5 million procedures annually. It is a dynamic market in
which we change paradigms by making non-invasive echotherapy the
new standard.
For more information, please visit www.theraclion.com and our
patient website www.echotherapy.com
Theraclion is listed on Euronext Growth Paris Mnemonic: ALTHE -
ISIN code: FR0010120402 LEI: 9695007X7HA7A1GCYD29
1 https://www.who.int/news-room/fact-sheets/detail/breast-cancer
2 Howlader N, Noone AM, Krapcho M, et al., eds. SEER Cancer
Statistics Review, 1975-2016. Bethesda, MD: National Cancer
Institute; 2019. Available from seer.cancer.gov/csr/1975_2016/,
based on November 2018 SEER data submission, posted to the SEER web
site April 2019
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220308006269/en/
David Auregan Chief Operating Officer
david.auregan@theraclion.com
Anja Kleber VP Marketing, Market Access & Sales Francophonia
anja.kleber@theraclion.com
Theraclion (EU:ALTHE)
Graphique Historique de l'Action
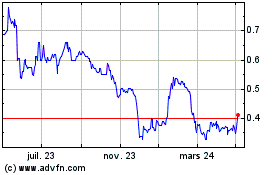
De Mar 2024 à Avr 2024
Theraclion (EU:ALTHE)
Graphique Historique de l'Action
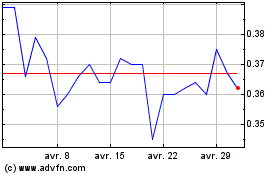
De Avr 2023 à Avr 2024