MedinCell Applauds Teva and Royalty Pharma Collaboration to Further Accelerate Olanzapine LAI Program (Codename: mdc-TJK or TEV-‘749)
13 Novembre 2023 - 3:38PM
Business Wire
- MedinCell’s partner Teva to receive R&D funding from
Royalty Pharma of up to $125 million to further accelerate the
development of olanzapine LAI, a long-acting subcutaneous
injectable olanzapine for schizophrenia using MedinCell’s
technology. (Access here Teva and Royalty Pharma today’s joint
press release for more details).
- Founded in 1996, Royalty Pharma is the largest buyer of
biopharmaceutical royalties and a leading funder of innovation
across the biopharmaceutical industry. Its current portfolio
includes royalties on over 35 products, many of which are
blockbusters, and it has 12 development-stage product
candidates.
- mdc-TJK is a once-monthly subcutaneous long-acting injection
of the atypical antipsychotic olanzapine that is currently in
clinical Phase 3 for the treatment of schizophrenia. It has the
potential to be the first long-acting olanzapine with a favorable
safety profile.
- Data of the Phase 3 trial are now expected in the second
half of 2024 (moved forward from 2025), as recently announced by
Teva (Q3 2023 earnings call, November 8, 2023 - Replay and a full
transcript available here)
- Following UZEDY™ (risperidone), approved by the FDA for the
treatment of schizophrenia in adults in April 2023, mdc-TJK is the
second product developed by Teva to utilize MedinCell’s proprietary
long-acting injectable technology (licensed to Teva under the name
SteadyTeq™).
- Teva remains fully responsible to lead the development and
commercialization of olanzapine LAI globally.
- MedinCell may receive up to $117 million in development and
commercial milestones over the coming years for mdc-TJK, and is
eligible for royalties on all net sales. The funding agreement
between Teva and Royalty Pharma does not affect future revenues to
which MedinCell may be entitled.
Regulatory News:
MedinCell (Paris:MEDCL):
In a joint press release announcing the financing agreement:
- Richard Francis, President and CEO of Teva said: “Since
launching Teva’s Pivot to Growth strategy in May 2023, we have been
working on novel and expedited ways to both continue to invest in
our robust innovative pipeline while supporting the growth of our
in-line businesses. We are excited to collaborate with Royalty
Pharma, a major funder of innovation with a strong track record,
experience, and reputation. This funding agreement enables us to
continue to accelerate the development of olanzapine LAI, a
critical program for us, without impacting resources dedicated to
our innovative and generic medicines.”
- Pablo Legorreta, founder and Chief Executive Officer of Royalty
Pharma said: "We are delighted to partner with Teva, to realize the
potential of olanzapine LAI and support them as their innovative
pipeline continues to come to fruition. Long-acting injectable
antipsychotics improve compliance and can help prevent
hospitalizations. Teva’s olanzapine LAI could be an important new
treatment option for patients with schizophrenia.”
About MedinCell
MedinCell is a clinical- and commercial-stage biopharmaceutical
company developing long-acting injectable drugs in many therapeutic
areas. Our innovative treatments aim to guarantee compliance with
medical prescriptions, to improve the effectiveness and
accessibility of medicines, and to reduce their environmental
footprint. They combine already known and used active ingredients
with our proprietary BEPO® technology which controls the delivery
of a drug at a therapeutic level for several days, weeks or months
from the subcutaneous or local injection of a simple deposit of a
few millimeters, entirely bioresorbable. The first treatment based
on BEPO technology, intended for the treatment of schizophrenia,
was approved by the FDA in April 2023, and is now distributed in
the United States by Teva under the name UZEDY™ (BEPO technology is
licensed to Teva under the name SteadyTeq™).
We collaborate with leading pharmaceutical companies and
foundations to improve global health through new treatment options.
Based in Montpellier, MedinCell currently employs more than 140
people representing more than 25 different nationalities.
UZEDY™ and SteadyTeq™ are trademarks of Teva Pharmaceuticals
www.medincell.com
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231113643382/en/
David Heuzé - Head of Communications
david.heuze@medincell.com / +33 (0)6 83 25 21 86
Media Relations Nicolas Mérigeau
medincell@newcap.eu / +33 (0)1 44 71 94 94
Investor Relations France Louis-Victor
Delouvrier/Alban Dufumier medincell@newcap.eu / +33 (0)1 44 71
94 94
Head of US Financial Strategy & IR Grace Kim
grace.kim@medincell.com / +1 (646) 991-4023
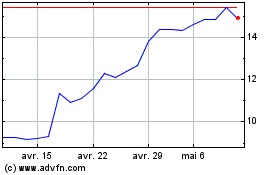
Medincell (EU:MEDCL)
Graphique Historique de l'Action
De Mai 2024 à Juin 2024
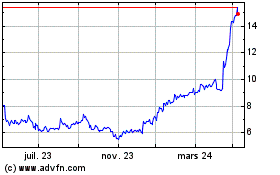
Medincell (EU:MEDCL)
Graphique Historique de l'Action
De Juin 2023 à Juin 2024