0001590560
false
00-0000000
0001590560
2023-08-01
2023-08-01
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of
the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
August 1, 2023
uniQure N.V.
(Exact Name of Registrant as Specified in
Charter)
| The Netherlands |
|
001-36294 |
|
N/A |
(State or Other
Jurisdiction of Incorporation) |
|
(Commission
File Number) |
|
(IRS Employer
Identification No.) |
Paasheuvelweg 25a,
1105 BP Amsterdam, The Netherlands |
|
N/A |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant’s telephone number, including
area code: +31-20-566-7394
(Former Name or Former Address, if Changed
Since Last Report)
Check the appropriate box below if the
Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions
(see General Instruction A.2. below):
¨ Written communications
pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
¨ Soliciting material
pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
¨ Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
¨ Pre-commencement communications pursuant to Rule 13e-4(c) under
the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class: |
|
Trading Symbol(s) |
|
Name of each exchange on which
registered: |
Ordinary
Shares, par value €0.05 per share |
|
QURE |
|
The Nasdaq Stock Market LLC
The Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging
growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of
the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the
registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards
provided pursuant to Section 13(a) of the Exchange Act. ¨
| Item 2.02 |
Results of Operations and Financial Condition |
On August 1, 2023, uniQure N.V. (the “Company”)
issued a press release announcing its financial results for the quarter ended June 30, 2023, and highlighting company progress. A
copy of the press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K.
The information furnished pursuant to this Item
2.02, including Exhibit 99.1, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange
Act of 1934, as amended (the “Exchange Act”) or otherwise subject to the liabilities under that Section and shall not
be deemed to be incorporated by reference into any filing of the Company under the Securities Act of 1933, as amended, or the Exchange
Act, except as shall be expressly set forth by specific reference in such filing.
| Item 9.01 |
Financial Statements and Exhibits |
(d) Exhibit
SIGNATURES
Pursuant to the requirements
of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto
duly authorized.
| |
UNIQURE N.V. |
| |
|
|
| Date: August 1, 2023 |
By: |
/S/ JEANNETTE POTTS |
| |
|
Jeannette Potts |
| |
|
Chief Legal and Compliance Officer |
Exhibit 99.1

uniQure Announces
Second Quarter 2023 Financial Results
and Highlights
Recent Company Progress
~ Announced encouraging interim analysis data
from ongoing Phase I/II clinical trial of AMT-130 in Huntington’s disease showing preservation of function compared to baseline
and clinical benefits relative to natural history; both dose cohorts of AMT-130 continue to be generally well-tolerated ~
~ Preparations
underway to initiate two Phase I/II clinical trials in refractory temporal lobe epilepsy and SOD1-ALS later this year; IND for Fabry
disease expected to be submitted in the second half of 2023 ~
~ Strong cash position of $628.6 million as
of June 30, 2023, which excludes a $100 million milestone related to first commercial sale of HEMGENIX® in the U.S.
received in July 2023 ~
~ Announced sale of royalty interest in HEMGENIX®
for up to $400 million ~
~ Appointed Walid Abi-Saab, M.D., as Chief Medical
Officer; Jeannette Potts as Chief Legal Officer ~
Lexington, MA and Amsterdam, the Netherlands,
August 1, 2023 — uniQure N.V. (NASDAQ: QURE), a leading gene therapy company
advancing transformative therapies for patients with severe medical needs, today reported its financial results for the second quarter
of 2023 and highlighted recent progress across its business.
“In the second quarter of the year, we
continued to make strong progress advancing our programs, including announcing encouraging data from the interim analysis of our Phase
I/II study of AMT-130 in Huntington’s disease,” stated Matt Kapusta, chief executive officer of uniQure. “These
data show that patients treated with AMT-130 appear to have generally preserved clinical and motor function and neurofilament light chain
(NfL) levels suggesting a stable to improving neurodegenerative profile. We believe that, collectively, the data suggest early and promising
evidence of clinical benefit in patients treated with AMT-130 compared to a patient-matched natural history data set. We look forward
to providing another update in the fourth quarter of 2023 that will include data from our European study, as well as preparing for regulatory
interactions in the first quarter of 2024 to discuss the data and potential late-stage development of AMT-130.”
“We are also working towards the initiation
of clinical trials for our gene therapy candidates in refractory temporal lobe epilepsy and SOD1-ALS later this year, as well as the submission
of an IND for our Fabry disease gene therapy,” he added. “With a strong cash position following achievement of the $100 million
milestone for first U.S. commercial sale of HEMGENIX® and completion of a royalty financing transaction, we are
focused on rapidly moving programs into clinical development and creating near-term value to shareholders.”
Recent Updates
| |
· | Advancing AMT-130 for the treatment of Huntington’s
disease |
| · | In June 2023, interim clinical
data were announced on the high-dose and low-dose cohorts of the double-blinded and randomized U.S. Phase I/II study of AMT-130 for the
treatment of early-stage Huntington’s disease. After 12- and 24-months
of follow-up, AMT-130 was generally well-tolerated with a manageable safety profile at both doses. For patients in both dose cohorts,
clinical function was generally preserved, and compared to natural history, demonstrated benefits at up to 24 months in each of Total
Motor Score, Total Functional Capacity and the composite Unified Huntington’s Disease Rating Scale. CSF NfL for the low-dose cohort
were below baseline at 24 months and were declining towards baseline for the high-dose cohort at 12 months. Suppression of CSF mHTT in
the low-dose cohort during 24 months of follow-up support target engagement, with greater variability observed in the high-dose cohort
during 12 months of follow-up. |
| · | Four control patients in the
high-dose cohort of the U.S. study have now crossed over to treatment with AMT-130. Three of these four patients received the high dose
of AMT-130, with one receiving the low dose. The remaining two control patients in the high-dose cohort did not meet the eligibility criteria
for crossover. All crossover patients received a short course of immunosuppression
therapy concurrent with the administration of AMT-130. |
| · | All six patients have been enrolled in the low-dose
cohort of the European, open-label Phase I/II clinical trial of AMT-130. The Company continues to make progress enrolling the second,
high-dose cohort of nine patients and expects to complete enrollment in the third quarter of 2023. |
| · | In the second half of 2023, uniQure expects to
initiate a third cohort in the ongoing U.S. clinical trial to further investigate both doses in combination with perioperative immunosuppression
with a focus on evaluating near-term safety and tolerability. The third cohort will enroll up to 10 patients, all of whom will receive
AMT-130 using the current, established stereotactic neurosurgical delivery procedure. |
| · | In the fourth quarter of 2023, the Company plans
to provide a clinical update from the U.S. and European Phase I/II studies of AMT-130, including 18- and 30-month follow-up data from
the treated patients in the high- and low-dose U.S. cohorts, respectively. The data update will include safety
and tolerability, biomarker and functional data across both high and low dose cohorts. |
| · | In the first quarter of 2024, the Company plans
on having regulatory interactions to discuss the U.S. and EU data and potential late-stage development pathways for AMT-130. |
| |
· | Expanding the Clinical Pipeline &
Advancing Innovation |
| · | AMT-260 for the treatment of refractory temporal
lobe epilepsy (rTLE) – In the first half of 2023, the Company completed its GLP toxicology study of AMT-260 in non-human primates.
The Company continues to expect submission of an investigational new drug (IND) application and initiation of a Phase I/II clinical study
of AMT-260 in the second half of 2023. |
| · | AMT-162 for the treatment of SOD1 amyotrophic
lateral sclerosis (ALS) - In January 2023, the Company entered into a global licensing agreement with Apic Bio for ABP-102, now
AMT-162 for the treatment of superoxide dismutase 1 (SOD1) amyotrophic lateral sclerosis (ALS), a rare, genetic form of ALS. The Company
expects to initiate a Phase I/II clinical study of AMT-162 in the second half of 2023. |
| · | AMT-191 for the treatment of Fabry disease
– In the third quarter of 2022, the Company initiated a GLP toxicology study of AMT-191 in non-human primates which is expected
to support an IND submission in the second half of 2023. |
| · | American Society of Gene and Cell Therapy
(ASGCT) 2023 – The company had a major presence at the annual ASGCT meeting held in Los Angeles, CA from May 16-20, 2023.
Twelve abstracts were accepted for presentation, including two oral presentations, that featured innovation in expanding the Company’s
research pipeline and advancements in AAV gene-therapy manufacturing. |
| |
· | Commercial launch of HEMGENIX®
(etranacogene dezaparvovec-drbl) for the treatment of hemophilia B in the United States and European Union |
| · | In June 2023, uniQure announced the achievement
of a $100 million milestone payment from CSL Behring associated with the first commercial sale of HEMGENIX® in the United
States. |
| · | In May 2023, uniQure announced the sale
of a royalty interest in HEMGENIX® for up to $400 million. The company received a $375 million upfront cash payment in
exchange for the lowest royalty tier on CSL Behring’s worldwide net sales of HEMGENIX® up to 1.85 times the
purchase price until June 30, 2032 or, if such cap is not met by June 30, 2032, up to 2.25x the purchase price
through December 31, 2038. uniQure will retain the rights to all other royalties under its existing Commercialization and License
Agreement with CSL Behring, as well as contractual milestones totaling up to $1.4 billion. |
| |
· | Strengthening of Leadership Team |
| · | In June 2023, Walid Abi-Saab, M.D., was
appointed as Chief Medical Officer, responsible for leading all clinical research and development, regulatory affairs, medical affairs,
and program management at uniQure. Dr. Abi-Saab brings more than 20 years of experience to the company having previously served as
Chief Medical Officer of Galapagos. Prior to joining Galapagos, he served as Group Vice President, Global Clinical Development at
Shire, where he was responsible for the clinical development plans of all programs in the therapeutic area and oversaw the development
of more than 10 marketed products and other programs in rare diseases and specialty pharma. He also has previously held leadership positions
at Novartis, Abbott Laboratories, and Pfizer. |
| · | In May 2023, Jeannette Potts, Ph.D. was
appointed as Chief Legal Officer, following the planned departure of David Cerveny. Ms. Potts has more than 25 years of global legal
experience in the biopharmaceutical industry. Most recently, she served as Senior Vice President, General Counsel & Corporate
Secretary at Forma Therapeutics, a clinical-stage biopharmaceutical company focused on rare hematologic diseases and cancer, which was
acquired by Novo Nordisk in 2022. Prior to joining Forma, Jeannette served as Head Counsel, Research and Development, at Takeda Pharmaceuticals,
and had previously been Vice President, Associate General Counsel at Millennium Pharmaceuticals before it was acquired by Takeda. |
| · | Strong cash position to advance the Company’s
programs |
| · | As of June 30, 2023, the
Company had cash, cash equivalents and investment securities of $628.6 million, which excludes the $100 million milestone payment received
from CSL Behring in July. The Company expects cash, cash equivalents and investment securities will fund operations into the second quarter
of 2026. |
Upcoming Investor Events
| |
· | Goldman Sachs Boston Bus Tour, August 9, 2023 – Boston, MA |
| |
· | Wells Fargo 2023 Healthcare Conference, September 6, 2023 – Boston,
MA |
| |
· | Citi’s 2023 18th Annual BioPharma Conference, September 7,
2023 – Boston, MA |
| |
· | 2023 Cantor Global Healthcare Conference, September 28, 2023 –
New York, NY |
Financial Highlights
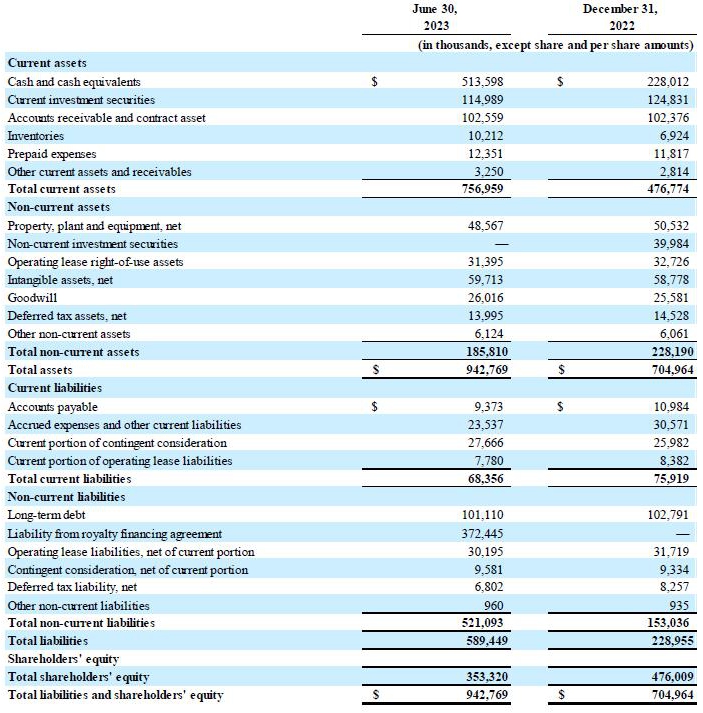
Cash position: As of June 30, 2023,
the Company held cash and cash equivalents and investment securities of $628.6 million, compared to $392.8 million as of December 31,
2022. The Company entered into a royalty agreement in May 2023 and received an upfront payment of $375.0 million.
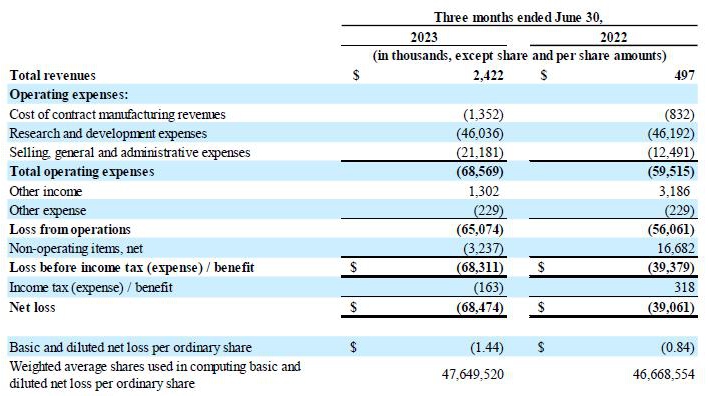
Revenues: Revenue for the three months
ended June 30, 2023 was $2.4 million, compared to $0.5 million in the same period in 2022. The increase is primarily a result of
contract manufacturing revenues of $1.3 million recognized in the current period related to contract manufacturing HEMGENIX®
for CSL Behring and an increase in license revenues of $0.8 million.
R&D expenses: Research and development
expenses were $46.0 million for the three months ended June 30, 2023, compared to $46.2 million in the same period in 2022. The decrease
in external program expense was offset by an increase in various other expenses, including employee and contractor-related expenses, facility
expenses and research and development disposable costs.
SG&A expenses: Selling, general and
administrative expenses were $21.2 million for the three months ended June 30, 2023, compared to $12.5 million in the same period
in 2022. The increase was primarily related to an increase in financial advisory fees, an increase in various other professional fees
and an increase in personnel and contractor-related expenses.
Other non-operating items, net:
Other non-operating items net was an expense of
$3.2 million for the three months ended June 30, 2023, compared to net income of $16.7 million for the same period in 2022. The decrease
in other non-operating items, net was primarily related to a decrease in foreign currency gains of $19.0 million and an increase in interest
expense.
Net loss:
The net loss for the three months ended June 30,
2023, was $68.5 million, or $1.44 basic and diluted loss per ordinary share, compared to $39.1 million net loss for the same period in
2022, or $0.84 basic and diluted loss per ordinary share.
About HEMGENIX®
HEMGENIX® is a gene therapy that
reduces the rate of abnormal bleeding in eligible people with hemophilia B by enabling the body to continuously produce factor IX, the
deficient protein in hemophilia B. It uses AAV5, a non-infectious viral vector, called an adeno-associated virus (AAV). The AAV5 vector
carries the Padua gene variant of Factor IX (FIX-Padua) to the target cells in the liver, generating factor IX proteins that are 5x-8x
more active than normal. These genetic instructions remain in the target cells, but generally do not become a part of a person’s
own DNA. Once delivered, the new genetic instructions allow the cellular machinery to produce stable levels of factor IX.
HEMGENIX® is a registered trademark
of CSL Behring.
About uniQure
uniQure is delivering on the promise of gene
therapy – single treatments with potentially curative results. The recent approvals of our gene therapy for hemophilia B –
an historic achievement based on more than a decade of research and clinical development – represent a major milestone in the field
of genomic medicine and ushers in a new treatment approach for patients living with hemophilia. We are now leveraging our modular and
validated technology platform to advance a pipeline of proprietary gene therapies for the treatment of patients with Huntington's
disease, refractory temporal lobe epilepsy, ALS, Fabry disease, and other severe diseases. www.uniQure.com
uniQure Forward-Looking Statements
This press release contains forward-looking
statements. All statements other than statements of historical fact are forward-looking statements, which are often indicated by terms
such as "anticipate," "believe," "could," “establish,” "estimate," "expect,"
"goal," "intend," "look forward to", "may," "plan," "potential," "predict,"
"project," “seek,” "should," "will," "would" and similar expressions. Forward-looking
statements are based on management's beliefs and assumptions and on information available to management only as of the date of this press
release. These forward-looking statements include, but are not limited to, the Company’s ability to fund operations into 2026, whether
there will be continued progress on the commercialization of HEMGENIX® and the Company’s ability to meet certain
milestones, statements about the clinical benefits for patients treated with AMT-130, the timing of patient enrollment in our AMT-130
clinical trial for Huntington’s Disease, whether the Company will announce follow up data from the U.S. Phase I/II clinical study
of AMT-130 and whether such announcement will include data from the Company’s European study, whether that data will help to further
guide our ongoing clinical development of AMT-130, whether the Company will submit an investigational new drug application for Fabry disease
in 2023, whether the company will initiate a Phase I/II clinical study of AMT-162 for the treatment of ALS in the second half of 2023,
whether the Company will submit an investigational new drug application or initiate a Phase I/II clinical study of AMT-260 for rTLE in
the second half of 2023, and whether the Company will begin interactions with regulatory agencies in the first quarter of 2024. The Company’s
actual results could differ materially from those anticipated in these forward-looking statements for many reasons, including, without
limitation, risks associated with the regulatory approval and commercial launch of HEMGENIX®, material changes to our interim
or preliminary data, our clinical trial for Huntington’s disease, the impact of financial and geopolitical events on our Company
and the wider economy and health care system, our Commercialization and License Agreement with CSL Behring, our clinical development activities,
clinical results, collaboration arrangements, regulatory oversight, product commercialization and intellectual property claims and ongoing
litigation, the as well as the risks, uncertainties and other factors described under the heading "Risk Factors" in the Company’s
periodic securities filings, including its Annual Report on Form 10-K filed February 27, 2023 and the Quarterly Report on Form 10-Q
filed August 1, 2023. Given these risks, uncertainties and other factors, you should not place undue reliance on these forward-looking
statements, and the Company assumes no obligation to update these forward-looking statements, even if new information becomes available
in the future.
uniQure Contacts:
| FOR INVESTORS: |
|
FOR MEDIA: |
| |
| Maria E. Cantor |
Chiara Russo |
Tom Malone |
| Direct: 339-970-7536 |
Direct: 617-306-9137 |
Direct: 339-970-7558 |
| Mobile: 617-680-9452 |
Mobile: 617-306-9137 |
Mobile:339-223-8541 |
| m.cantor@uniQure.com |
c.russo@uniQure.com |
t.malone@uniQure.com |
uniQure N.V.
UNAUDITED CONSOLIDATED BALANCE SHEETS
uniQure N.V.
UNAUDITED CONSOLIDATED STATEMENTS OF OPERATIONS
v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionISO 3166-1 alpha-2 country code.
| Name: |
dei_EntityAddressCountry |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:countryCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
uniQure NV (NASDAQ:QURE)
Graphique Historique de l'Action
De Août 2024 à Sept 2024

uniQure NV (NASDAQ:QURE)
Graphique Historique de l'Action
De Sept 2023 à Sept 2024
