Teva, MedinCell Say FDA Accepts NDA for Schizophrenia Treatment
31 Août 2021 - 11:11PM
Dow Jones News
By Stephen Nakrosis
Teva Pharmaceuticals and MedinCell on Tuesday said the U.S. Food
and Drug Administration accepted their new drug application for
TV-46000/mdc-IRM to treat schizophrenia.
The companies said the acceptance of TV-46000/mdc-IRM, or
risperidone extended-release injectable suspension for subcutaneous
use, was based on Phase 3 data from two studies which "evaluated
the efficacy and long-term safety and tolerability of TV-46000 as a
treatment for patients with schizophrenia." The companies said the
results will be shared "at future scientific conferences and in
peer-reviewed publications."
Teva will continue to lead the clinical development and
regulatory process and will be responsible for commercialization,
while MedinCell will be eligible for certain milestones and
royalties, the companies said.
Teva Pharmaceuticals is a U.S. affiliate of Teva Pharmaceutical
Industries Ltd.
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
August 31, 2021 16:56 ET (20:56 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
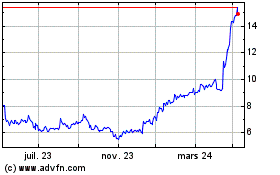
Medincell (EU:MEDCL)
Graphique Historique de l'Action
De Avr 2024 à Mai 2024
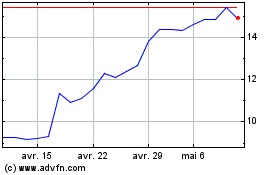
Medincell (EU:MEDCL)
Graphique Historique de l'Action
De Mai 2023 à Mai 2024