Alvotech Gets FDA Letter, Expects Delay in U.S. Approval of AVT02
29 Juin 2023 - 2:03AM
Dow Jones News
By Mary de Wet
Alvotech said Wednesday that approval for AVT02, a biosimilar
candidate for Abbvie's Humira, in the U.S. is likely to be delayed
after the Icelandic company received a complete response letter
from the Food and Drug Administration.
As a result, Alvotech said it would explore options to raise
additional capital to continue advancing pipeline development in
the near term.
ATP Holdings, a unit of Alvotech's largest shareholder, Aztiq,
is interested in providing as much as $100 million from potential
financing, Alvotech said.
The FDA letter was in response to the company's second biologics
license application. In April, the FDA sent Alvotech a complete
response letter for its initial application, which noted certain
deficiencies at the company's Reykjavik facility.
Alvotech said it intends to resubmit the biologics license
application for AVT02.
Write to Mary de Wet at mary.dewet@dowjones.com
(END) Dow Jones Newswires
June 28, 2023 19:48 ET (23:48 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
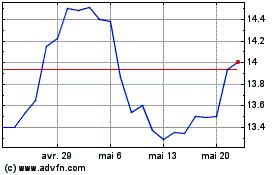
Alvontech (NASDAQ:ALVO)
Graphique Historique de l'Action
De Avr 2024 à Mai 2024
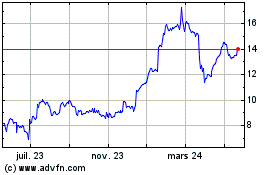
Alvontech (NASDAQ:ALVO)
Graphique Historique de l'Action
De Mai 2023 à Mai 2024