Optinose Shares Slide Premarket as FDA Extends Xhance Review
06 Décembre 2023 - 2:48PM
Dow Jones News
By Colin Kellaher
Shares of Optinose plunge in premarket trading Wednesday after
the pharmaceutical company said the U.S. Food and Drug
Administration has extended the review of a request for expanded
approval of its Xhance nasal spray by three months.
Optinose, which is seeking approval of Xhance as a treatment for
chronic rhinosinusitis, said the FDA needs more time to review
additional efficacy subset analyses the Yardley, Pa., company
recently submitted at the agency's request.
Optinose said the FDA extended its target action date on the
application to March 16, 2024.
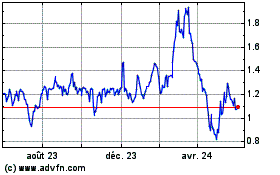
Optinose shares, which closed Tuesday at $1.47, were recently
down 22% to $1.14 in premarket trading.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
December 06, 2023 08:33 ET (13:33 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
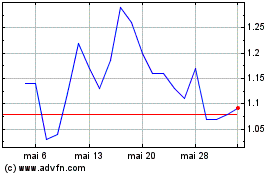
OptiNose (NASDAQ:OPTN)
Graphique Historique de l'Action
De Mai 2024 à Juin 2024
OptiNose (NASDAQ:OPTN)
Graphique Historique de l'Action
De Juin 2023 à Juin 2024