Oramed has completed an analysis of its U.S.-based Phase 3
oral insulin trial with significant lowering of A1C levels seen in
patient subgroups
NEW
YORK, May 15, 2023 /PRNewswire/ -- Oramed
Pharmaceuticals Inc. (Nasdaq: ORMP) (TASE: ORMP) (www.oramed.com)
announced today that Hefei Tianhui Biotechnology Co. Ltd.
(HTIT), a strategic partner of Oramed, has successfully completed
its Phase 3 trials of oral insulin in type 2 diabetes in
China under a differentiated study
protocol. HTIT is now moving toward regulatory approval and
has submitted the data to the National Medical Products
Administration (NMPA, formerly the CFDA).
Oramed has recently completed an analysis of the data from its
U.S.-based, Phase 3 trial, ORA-D-013-1, for the treatment of type 2
diabetes. This analysis found that subpopulations of patients
with pooled specific parameters, such as body mass index
(BMI), baseline HbA1c, age, gender and body
weight, responded well to oral insulin. These subsets
exhibited an over 1% placebo adjusted, statistically significant,
reduction in HbA1c. The significant impact of baseline BMI on
the responder group within the U.S. Phase 3 trial aligns with
the positive data from the HTIT trial in China. The U.S.
subpopulation and the Chinese general trial population shared a
very similar baseline BMI.
"We are excited by our partner, HTIT's, success and share in
their excitement as they move one step closer to commercialization
in China," said Oramed Chief
Executive Officer, Nadav Kidron.
"Additionally, we are encouraged by the review of our Phase 3 data
which found a strong correlation between certain parameters and the
oral insulin's efficacy. Based on these findings, Oramed is
exploring ways to move forward with its oral insulin product," Mr.
Kidron added.
About Oramed Pharmaceuticals
Oramed Pharmaceuticals (Nasdaq: ORMP) (TASE: ORMP) is a platform
technology pioneer in the field of oral delivery solutions for
drugs currently delivered via injection. The Company's novel
Protein Oral Delivery (POD™) technology is designed to protect drug
integrity and increase absorption. Oramed has offices in
the United States and Israel. For more information, please
visit www.oramed.com.
About the Methodology Used for US Phase 3 Data
Analysis
Oramed applied two separate methodologies to retrospectively
analyze the U.S. Phase 3 data*.
1. The Company applied causal-machine learning (ML) analysis to
identify potential subgroup responders**. This method was
successful in identifying multiple features responsible for the
causal treatment effects. The two most important features were BMI
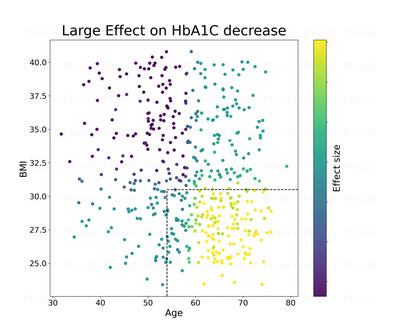
and age as can be seen in the chart to the right.
Legend: X axis represents the age of patients in the trial
Y axis represents the BMI of patients in the trial
Bright colors
(yellowish) represent large reduction in A1C for the treatment
patients vs. placebo
Conclusion: Patients over 54 of age with BMI of less than 31
tend to have the largest reduction in A1c p-value lower than
0.002.
2. The analysis included a search for markers (demographic and
clinical) that demonstrate the highest A1c improvement (measured
between baseline and Visit 6) for each of the treatments compared
with placebo for men, women, and both***. The results are
displayed in the chart to the right.
* The data was compiled from 489 patients who completed both
visit 6 (at 24 weeks) and all required laboratory data.
** Analysis was performed by PhaseV (https://phasevtrials.com/)
*** Analysis was performed by Panacea
(https://www.panacea-ml.com/)
About HTIT
Hefei Tianhui Biotechnology Co., Ltd. (HTIT), has a
state-of-the-art oral insulin manufacturing facility in
Hefei, China. HTIT is a
high-tech company focused on biopharmaceutical product
manufacturing and R&D with an emphasis on the oral delivery of
therapeutic macromolecules.
Forward-looking statements: This press release
contains forward-looking statements. For example, we are using
forward-looking statements when we discuss potential marketing
approval by HTIT and commercialization in China, the potential safety and efficacy of
oral insulin and the potential of Oramed to move forward with
its oral insulin product. These forward-looking statements are
based on the current expectations of the management of Oramed only,
and are subject to a number of factors and uncertainties that could
cause actual results to differ materially from those described in
the forward-looking statements, including the Company's process to
evaluate strategic options; the terms, timing, structure, benefits
and costs of any strategic transaction and whether any transaction
will be consummated at all; the impact of any strategic transaction
on the Company; the outcomes of any litigation, regulatory
proceedings, inquiries or investigations to which the Company may
be subject; the ability to obtain financing or third-party
approvals as needed; our ability to achieve the intended benefits
of our strategic initiatives; the risks and uncertainties related
to the progress, timing, cost, and results of clinical trials and
product development programs; difficulties or delays in obtaining
regulatory approval or patent protection for our product
candidates; competition from other pharmaceutical or biotechnology
companies; and our ability to obtain additional funding required to
conduct our research, development and commercialization activities.
In addition, the following factors, among others, could cause
actual results to differ materially from those described in the
forward-looking statements: changes in technology and market
requirements; delays or obstacles in launching our clinical trials;
changes in legislation; inability to timely develop and introduce
new technologies, products and applications; lack of validation of
our technology as we progress further and lack of acceptance of our
methods by the scientific community; inability to retain or attract
key employees whose knowledge is essential to the development of
our products; unforeseen scientific difficulties that may develop
with our process; greater cost of final product than anticipated;
loss of market share and pressure on pricing resulting from
competition; laboratory results that do not translate to equally
good results in real settings; our patents may not be sufficient;
that products may harm recipients; and other factors discussed in
the "Risk Factors" section of the Company's most recent Annual
Report on Form 10-K and Quarterly Reports on Form 10-Q, which are
on file with the Securities and Exchange Commission and in other
filings that the Company makes with the Securities and Exchange
Commission in the future. All of these factors could cause the
actual results or performance of Oramed to differ materially from
those contemplated in such forward-looking statements. Except as
otherwise required by law, Oramed undertakes no obligation to
publicly release any revisions to these forward-looking statements
to reflect events or circumstances after the date hereof or to
reflect the occurrence of unanticipated events.
Company Contact:
Zach Herschfus
+1-844-9-ORAMED
zach@oramed.com
Photo -
https://mma.prnewswire.com/media/2077037/Oramed_Pharmaceuticals.jpg
Photo -
https://mma.prnewswire.com/media/2077038/Oramed_Pharmaceuticals_2.jpg
Logo -
https://mma.prnewswire.com/media/2077043/Oramed_Logo.jpg
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/oramed-announces-that-its-chinese-partner-htit-has-successfully-completed-a-phase-3-oral-insulin-clinical-trial-and-submitted-a-marketing-authorization-application-in-china-301824604.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/oramed-announces-that-its-chinese-partner-htit-has-successfully-completed-a-phase-3-oral-insulin-clinical-trial-and-submitted-a-marketing-authorization-application-in-china-301824604.html
SOURCE Oramed Pharmaceuticals Inc.