PURATEIN(R) AND SUPERTEIN(TM) GRAS NOTIFIED - U.S. FOOD & DRUG ADMINISTRATION ISSUES NO OBJECTION LETTER ON GRAS STATUS OF PURAT
30 Août 2010 - 12:30PM
PR Newswire (Canada)
VANCOUVER, Aug. 30 /CNW/ -- VANCOUVER, Aug. 30 /CNW/ - Burcon
NutraScience Corporation (TSX - BU) ("Burcon") announced today that
the U.S. Food and Drug Administration (FDA or the Agency) has
issued a no objection letter with respect to Puratein(R) and
Supertein(TM) canola protein isolates. This response indicates the
FDA has no objection to the conclusion that Puratein(R) and
Supertein(TM) are Generally Recognized as Safe (GRAS) among
qualified experts for use alone or together as an ingredient in
dairy products, grain products, fruit and vegetable juices and
beverages, salad dressings, meal replacements, and nutritional
bars. Based on the information provided, as well as other
information available to the FDA, the Agency has no questions at
this time regarding the conclusion that Puratein(R) and
Supertein(TM) are GRAS under the intended conditions of use. As
such, Puratein(R) and Supertein(TM) are now considered to be GRAS
Notified. Burcon's patented process for the production of
Puratein(R) and Supertein(TM) canola protein isolates broadly
separates the two naturally occurring proteins found in canola. The
two naturally occurring proteins in canola, cruciferin and napin,
have highly distinct functional and nutritional characteristics,
similar to the way that the two proteins in egg, egg-yolk and
egg-white, have distinct characteristics. Burcon's patented process
for the production of Puratein(R) and Supertein(TM) canola protein
isolates thereby creates two distinct protein ingredients with
separate and distinct nutritional and functional profiles allowing
for their use in a broader variety of food and beverage
applications. "Receipt of the official response from the U.S. Food
& Drug Administration paves the way for the advancement of our
canola protein isolates Puratein(R) and Supertein(TM). Producers of
branded food and beverage products demand quality safe
ingredients," said Johann F. Tergesen, President and Chief
Operating Officer, adding, "To our knowledge, no other canola
protein has achieved this level of regulatory recognition. This
marks a significant milestone in the development of Puratein(R) and
Supertein(TM) canola protein isolates for use in food and beverage
applications." About Burcon NutraScience Burcon is a leader in
nutrition, health and wellness in the field of functional,
renewable plant proteins. Since 1999, Burcon has developed a
portfolio of composition, application, and process patents
originating from our core protein extraction and purification
technology. We are developing Puratein(R) and Supertein(TM) canola
protein isolates with unique functional and nutritional attributes,
and CLARISOY(R), a revolutionary soy protein isolate which is 100%
soluble and completely transparent in acidic solutions. Our team of
highly specialized scientists and engineers work from our own
research facility to develop and optimize environmentally sound
technologies. To-date, our patent portfolio consists of 135 issued
patents in various countries, including 20 issued U.S. patents, and
in excess of 200 additional pending patent applications, 66 of
which are U.S. patent applications. ON BEHALF OF THE BOARD OF
DIRECTORS "Johann F. Tergesen" Johann F. Tergesen President and
Chief Operating Officer The TSX has not reviewed and does not
accept responsibility for the adequacy of the content of the
information contained herein. This press release contains
forward-looking statements that involve risks and uncertainties.
These forward-looking statements relate to, among other things,
plans and timing for the introduction or enhancement of our
products, statements about future market conditions, supply and
demand conditions, and other expectations, intentions and plans
contained in this press release that are not historical fact. Our
expectations regarding the prospect for future success depend upon
our ability to develop and sell products, which we do not produce
today and cannot be sold without further research and development.
When used in this press release, the words "goal", "intend",
"believes" and "potential" and similar expressions, generally
identify forward-looking statements. These statements reflect our
current expectations. They are subject to a number of risks and
uncertainties. In light of the many risks and uncertainties
surrounding the development of a source of protein from canola
meal, you should understand that we cannot assure you that the
forward looking statements contained in this press release will be
realized. Jade Cheng, Chief Financial Officer, Burcon NutraScience
Corporation, (604) 733-0896, (888) 408-7960 toll-free,
jcheng@burcon.ca, www.burcon.ca; AXINO AG, Wolfgang Seybold,
Investor Relations Europe, AXINO AG, Konigstrasse 26, 70173
Stuttgart, Germany, Tel. +49-711-25 35 92-40, Fax +49-711-25 35
92-55, wolfgang.seybold@axino.de, www.burcon.net
Copyright
Burcon NutraScience (TSX:BU)
Graphique Historique de l'Action
De Mai 2024 à Juin 2024
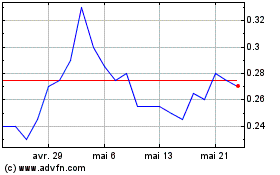
Burcon NutraScience (TSX:BU)
Graphique Historique de l'Action
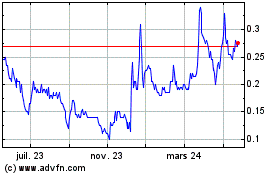
De Juin 2023 à Juin 2024