UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of November, 2023.
Commission File Number: 001-40673
Cybin Inc.
(Exact Name of Registrant as Specified in Charter)
100 King Street West, Suite 5600, Toronto, Ontario, M5X 1C9
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20-F or Form 40-F.
Form 20-F □ Form 40-F ⊠
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | | | | | | | |
| | CYBIN INC. | |
| | (Registrant) | |
| | | | |
| Date: | November 21, 2023 | By: | /s/ Doug Drysdale | |
| | Name: | Doug Drysdale | |
| | Title: | Chief Executive Officer | |
EXHIBIT INDEX
Cybin to Host CYB003 Topline Depression Study Review and R&D Briefing on November 30, 2023, in New York City
- Company speakers include Doug Drysdale, Chief Executive Officer; and Amir Inamdar, MBBS, DNB (Psych), MFPM, Chief Medical Officer -
- Featured key opinion leaders include Dr. Maurizio Fava, M.D., Massachusetts General Hospital and Harvard Medical School; and Dr. Gitte Moos Knudsen, Professor, Chief Neurologist, DMSc, Neurobiology Research Unit, Rigshospitalet and University of Copenhagen, Denmark -
- Topics include topline Phase 2 safety and efficacy data for CYB003, Cybin’s deuterated psilocybin analog being developed for the treatment of major depressive disorder, and a panel discussion with key opinion leaders on data and study designs, the current treatment landscape in depression, and the future role of psychedelics -
TORONTO, CANADA – November 20, 2023 – Cybin Inc. (NYSE American:CYBN) (NEO:CYBN) (“Cybin” or the “Company”), a clinical-stage biopharmaceutical company committed to revolutionizing mental healthcare by developing new and innovative psychedelic-based treatment options, today announced that it will discuss its topline Phase 2 safety and efficacy data for CYB003, its deuterated psilocybin analog being developed for the treatment of major depressive disorder, as part of an R&D investor briefing featuring a key opinion leader panel titled “Understanding the Current Treatment Landscape in Depression and Therapeutic Potential of Psychedelics in Mental Health” on Thursday, November 30, 2023 in New York City, at 10:00 a.m. ET.
Key Opinion Leaders
Dr. Maurizio Fava is Chair of the Department of Psychiatry/Psychiatrist-in-Chief of the Massachusetts General Hospital (“MGH”), executive director of the Clinical Trials Network and Institute, MGH, Associate Dean for clinical and translational research and the Slater Family Professor of Psychiatry at Harvard Medical School. Dr. Fava is a world leader in the field of depression and clinical psychopharmacology. He has edited eight books and authored or co-authored more than 900 original articles published in medical journals with international
circulation, articles which have been cited more than 100,000 times in the literature and with an h-index greater than 150 on Google Scholar.
Dr. Fava obtained his medical degree from the University of Padova School of Medicine and completed residency training in endocrinology at the same university. He then moved to the United States and completed residency training in psychiatry at MGH. He founded and was director of the hospital’s Depression Clinical and Research Program from 1990 until 2014. Under Dr. Fava’s direction, the Depression Clinical and Research Program became one of the most highly regarded depression programs in the country, a model for academic programs that link, in a bi-directional fashion, clinical and research work. In 2007, he also founded and is now the executive director of the MGH Psychiatry Clinical Trials Network and Institute, the first academic CRO specialized in the coordination of multi-center clinical trials in psychiatry. He is the former President of the American Society of Clinical Psychopharmacology and, in 2023, Dr. Fava was elected to the National Academy of Medicine.
Dr. Gitte M. Knudsen is Chair of Neurobiology Research Unit, Rigshospitalet and University of Copenhagen, Denmark, past president of the European College of Neuropsychopharmacology and chair of the advisory board for the EU flagship The Human Brain Project. Dr. Knudsen is a world leader in neuropharmacology and molecular and functional neuroimaging and leads investigator-initiated trials on psychedelics. As the principal investigator of BrainDrugs, a precision medicine research alliance, her work aims to identify which features can form a basis for decisions regarding the right treatment for the right patient with epilepsy or major depressive disorder. She has authored or co-authored 30 books/book chapters and 500 original articles published in medical journals with international circulation, articles which have been cited 25,000 times in the literature and with an h-index of 78 on Google Scholar.
Professor Knudsen received her medical and doctoral degrees from the University of Copenhagen, Denmark and completed her training in neurology. She has received numerous awards and honors, including e.g., the Monrad-Krohn Prize; Niels Lassen Prize and The Carlsberg Foundation Researcher Award for Sciences. She has been a member of the Royal Danish Academy of Sciences and Letters since 2004 and is appointed Adjunct Professor at the University of Vienna, Austria.
Program Schedule:
| | | | | |
Date: | Thursday, November 30, 2023 |
Presentations: | 10:00 a.m. - 10:45 a.m. ET |
Panel Discussion: | 10:45 a.m. - 11:15 a.m. ET |
Q&A: | 11:15 a.m. - 11:30 a.m. ET |
A live webcast of the event will be available on the Company’s investor relations website on the Events & Presentations page.
About Cybin
Cybin is a clinical-stage biopharmaceutical company on a mission to create safe and effective psychedelic-based therapeutics to address the large unmet need for new and innovative treatment options for people who suffer from mental health conditions.
Cybin’s goal of revolutionizing mental healthcare is supported by a network of world-class partners and internationally recognized scientists aimed at progressing proprietary drug discovery platforms, innovative drug delivery systems, and novel formulation approaches and treatment regimens. The Company is currently developing CYB003, a proprietary deuterated psilocybin analog for the treatment of major depressive disorder and CYB004, a proprietary deuterated DMT molecule for generalized anxiety disorder and has a research pipeline of investigational psychedelic-based compounds.
Headquartered in Canada and founded in 2019, Cybin is operational in Canada, the United States, the United Kingdom, the Netherlands and Ireland. For company updates and to learn more about Cybin, visit www.cybin.com or follow the team on X, LinkedIn, YouTube and Instagram.
Cautionary Notes and Forward-Looking Statements
Certain statements in this news release relating to the Company are forward-looking statements and are prospective in nature. Forward-looking statements are not based on historical facts, but rather on current expectations and projections about future events and are therefore subject to risks and uncertainties which could cause actual results to differ materially from the future results expressed or implied by the forward-looking statements. These statements generally can be identified by the use of forward-looking words such as “may”, “should”, “could”, “intend”, “estimate”, “plan”, “anticipate”, “expect”, “believe” or “continue”, or the negative thereof or similar variations. Forward-looking statements in this news release include statements regarding the Company’s plans to report Phase 2 safety and efficacy data from its CYB003 program in Q4 2023; and the Company’s plans to engineer proprietary drug discovery platforms, innovative drug delivery systems, novel formulation approaches and treatment regimens for mental health conditions.
These forward-looking statements are based on reasonable assumptions and estimates of management of the Company at the time such statements were made. Actual future results may differ materially as forward-looking statements involve known and unknown risks, uncertainties, and other factors which may cause the actual results, performance, or achievements of the Company to materially differ from any future results, performance, or achievements expressed or implied by such forward-looking statements. Such factors, among other things, include: implications of the spread of COVID-19 on the Company's operations; fluctuations in general macroeconomic conditions; fluctuations in securities markets; expectations regarding the size of the psychedelics market; the ability of the Company to successfully achieve its business objectives; plans for growth; political, social and environmental uncertainties; employee
relations; the presence of laws and regulations that may impose restrictions in the markets where the Company operates; and the risk factors set out in each of the Company's management's discussion and analysis for the three and six month periods ended September 30, 2023, and the Company’s annual information form for the year ended March 31, 2023, which are available under the Company's profile on SEDAR+ at www.sedarplus.ca and with the U.S. Securities and Exchange Commission on EDGAR at www.sec.gov. Although the forward-looking statements contained in this news release are based upon what management of the Company believes, or believed at the time, to be reasonable assumptions, the Company cannot assure shareholders that actual results will be consistent with such forward-looking statements, as there may be other factors that cause results not to be as anticipated, estimated or intended. Readers should not place undue reliance on the forward-looking statements and information contained in this news release. The Company assumes no obligation to update the forward-looking statements of beliefs, opinions, projections, or other factors, should they change, except as required by law.
Cybin makes no medical, treatment or health benefit claims about Cybin’s proposed products. The U.S. Food and Drug Administration, Health Canada or other similar regulatory authorities have not evaluated claims regarding psilocybin, psychedelic tryptamine, tryptamine derivatives or other psychedelic compounds. The efficacy of such products has not been confirmed by approved research. There is no assurance that the use of psilocybin, psychedelic tryptamine, tryptamine derivatives or other psychedelic compounds can diagnose, treat, cure or prevent any disease or condition. Rigorous scientific research and clinical trials are needed. Cybin has not conducted clinical trials for the use of its proposed products. Any references to quality, consistency, efficacy and safety of potential products do not imply that Cybin verified such in clinical trials or that Cybin will complete such trials. If Cybin cannot obtain the approvals or research necessary to commercialize its business, it may have a material adverse effect on Cybin’s performance and operations.
Neither the Neo Exchange Inc. nor the NYSE American LLC stock exchange have approved or disapproved the contents of this news release and are not responsible for the adequacy and accuracy of the contents herein.
Investor & Media Contact:
Gabriel Fahel
Chief Legal Officer
Cybin Inc.
1-866-292-4601
irteam@cybin.com – or – media@cybin.com
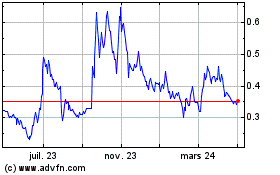
Cybin (AMEX:CYBN)
Graphique Historique de l'Action
De Juin 2024 à Juil 2024
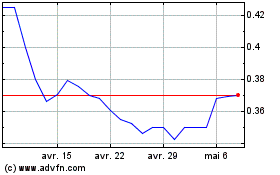
Cybin (AMEX:CYBN)
Graphique Historique de l'Action
De Juil 2023 à Juil 2024