Amicus Therapeutics Gets FDA Approval for Pombiliti + Opfolda
28 Septembre 2023 - 5:31PM
Dow Jones News
By Chris Wack
Trading in shares of Amicus Therapeutics was halted at $12.88 as
the Food and Drug Administration approved Pombiliti + Opfolda 65mg
capsules for adults living with late-onset Pompe disease, and who
aren't improving on their current enzyme replacement therapy.
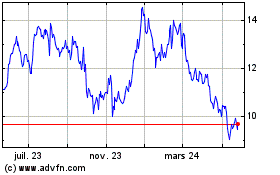
The stock hit its 52-week high of $14.10 on Aug. 9, and is up
20% in the past 12 months.
Late-onset Pompe disease is a rare, debilitating, and
life-threatening lysosomal disorder caused by a deficiency of the
enzyme acid alpha-glucosidase.
Pombiliti is a recombinant human enzyme naturally expressed with
high levels of bis-Mannose 6-Phosphate, designed for increased
uptake into muscle cells. Opfolda is an enzyme stabilizer designed
to stabilize the enzyme in the blood.
The FDA approval was based on clinical data observed from a
Phase 3 pivotal study, the only trial in late-onset Pompe disease
to study ERT-experienced participants in a controlled setting.
Amicus Therapeutics will launch Pombiliti + Opfolda immediately
in the U.S. The FDA previously granted Breakthrough Therapy
designation for Pombiliti + Opfolda. It has also been approved for
the treatment of adults with late-onset Pompe disease in the E.U.
and the U.K.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
September 28, 2023 11:16 ET (15:16 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
Amicus Therapeutics (NASDAQ:FOLD)
Graphique Historique de l'Action
De Mai 2024 à Juin 2024
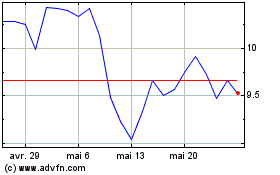
Amicus Therapeutics (NASDAQ:FOLD)
Graphique Historique de l'Action
De Juin 2023 à Juin 2024