- Disappearance of lesions and absence of residual
malignant cells in Luminal-A group considered to be predictive
for the complete effectiveness of the treatment
- This is the latest in a growing body of evidence
presented by independent, non-sponsored doctors who
use ProSense
CAESAREA, Israel, Oct. 5, 2023
/PRNewswire/ -- IceCure Medical Ltd. (Nasdaq: ICCM) ("IceCure"
or the "Company"), developer of the ProSense® System, a
minimally-invasive cryoablation technology that destroys tumors by
freezing as an alternative to surgical tumor removal, today
announced that the findings from an independent breast cancer
study, performed by a leading radiologist in Italy, support the safety and efficacy of the
Company's ProSense, a minimally-invasive cryoablation system. This
independent study adds to the growing body of scientific data using
ProSense in women deemed inoperable for breast cancer.
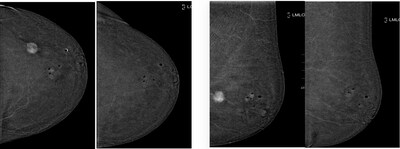
During the study, ultrasound-guided cryoablation using ProSense
was performed on 28 women, who had biopsy-proven malignant lesions,
and were deemed inoperable by a multidisciplinary group, and
submitted to hormone therapy. At a median age of 73.4 years, 14 of
the women were diagnosed with molecular subtype Luminal-A tumors
and another 14 diagnosed with Luminal-B tumors. Patients were
monitored at one, three, and six months post-procedure, at which
time the tumor size reduction rate was evaluated by ultrasound. The
effectiveness of the procedure was further evaluated after one year
by core needle biopsy on the post-procedural scar (inside the
breast at the site of the tumor) to determine the absence of
residual tumoral cells. The size reduction rates were as
follows:
1
month: 21.59%
for Luminal-A vs. 19.83% for Luminal-B
3 months: 70.16% for Luminal-A
vs. 60.71% for Luminal-B
6 months: 96.81% for Luminal-A
vs. 93.43% for Luminal-B
In the Luminal-A group, there was an absence of residual
malignant cells in all biopsy samples and a disappearance of
lesions, leading the study to conclude this is a predictive factor
for the complete effectives of treatment. Luminal-A is the most
common subtype and represents 50%-60% of all breast
cancers1.
"The patients in the study were deemed inoperable and had no
treatment options available other than hormone therapy, and as we
observe breast cancer awareness month, it is reassuring that a
minimally invasive cryoablation procedure using IceCure's ProSense
system is an available alternative," stated IceCure's CEO,
Eyal Shamir. "This was a relatively
short independent study that measured tumor size reduction, however
the highly favorable results nonetheless validate similar results
we are experiencing in our longer-term five-year post-procedure
ICE3 study, which we expect to conclude in the first quarter of
2024. We believe the increasing number of doctors using ProSense
and conducting these studies on their own initiative is the best
testament to the usability and benefit of ProSense in real-world
clinical settings."
The data from the Italy
study was presented in a poster titled, "Assessing the
outcome of cryoablation treatment on different molecular subtype of
low-grade breast cancer," at the European Society of Breast Imaging
("EUSOBI") Scientific Meeting that was recently held in
Valencia, Spain. The data was
presented by Principal Investigator, Dr. F.
Di Naro, of Azienda Ospedaliero-Universitaria Careggi,
Diagnostic Senology Unit, Florence,
Italy. ProSense is approved in Italy for numerous indications, including
breast cancer.
1. Yersal O, Barutca S. Biological subtypes of
breast cancer: Prognostic and therapeutic implications. World J
Clin Oncol. 2014 Aug 10;5(3):412-24. doi: 10.5306/wjco.v5.i3.412.
PMID: 25114856; PMCID: PMC4127612.
About IceCure Medical
IceCure Medical (Nasdaq: ICCM) develops and markets ProSense®,
an advanced liquid-nitrogen-based cryoablation therapy for the
treatment of tumors (benign and cancerous) by freezing, with the
primary focus areas being breast, kidney, bone and lung cancer. Its
minimally invasive technology is a safe and effective alternative
to hospital surgical tumor removal that is easily performed in a
relatively short procedure. The system is marketed and sold
worldwide for the indications cleared and approved to date
including in the U.S., Europe, and
China.
Forward Looking Statements
This press release contains forward-looking statements within
the meaning of the "safe harbor" provisions of the Private
Securities Litigation Reform Act of 1995 and other Federal and
Israeli securities laws. Words such as "expects," "anticipates,"
"intends," "plans," "believes," "seeks," "estimates" and similar
expressions or variations of such words are intended to identify
forward-looking statements. For example, IceCure is using forward
looking statement in this press release when it discusses: the
expected conclusion of its ICE3 study in the first quarter of 2024;
and the belief that the increasing number of doctors using ProSense
and conducting studies with ProSense is a testament to the
usability and benefit of ProSense in real-world clinical settings.
Historic results of scientific research, studies and clinical and
preclinical trials do not guarantee that the conclusions of future
research or trials will suggest identical or even similar
conclusions. Because such statements deal with future events and
are based on IceCure's current expectations, they are subject to
various risks and uncertainties and actual results, performance, or
achievements of IceCure could differ materially from those
described in or implied by the statements in this press release.
The forward-looking statements contained or implied in this press
release are subject to other risks and uncertainties, many of which
are beyond the control of the Company, including those set forth in
the Risk Factors section of the Company's Annual Report on Form
20-F for the year ended December 31,
2022 filed with the SEC on March 29,
2023, and other documents filed with or furnished
to the SEC which are available on the SEC's website,
www.sec.gov. The Company undertakes no obligation to update these
statements for revisions or changes after the date of this release,
except as required by law.
IR Contact:
Email: investors@icecure-medical.com
Michael Polyviou
Phone: 732-232-6914
Todd Kehrli
Phone: 310-625-4462
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/women-deemed-inoperable-for-breast-cancer-benefitted-from-icecure-medicals-prosense-as-an-independent-study-performed-in-italy-showed-a-tumor-reduction-rate-of-93-43-to-96-81-301948271.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/women-deemed-inoperable-for-breast-cancer-benefitted-from-icecure-medicals-prosense-as-an-independent-study-performed-in-italy-showed-a-tumor-reduction-rate-of-93-43-to-96-81-301948271.html
SOURCE IceCure Medical Ltd