0000714256
false
0000714256
2023-08-14
2023-08-14
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
8-K
CURRENT
REPORT
Pursuant
to Section 13 or 15(d) of the
Securities
Exchange Act of 1934
Date
of Report (Date of earliest event reported): August 14, 2023
| SANARA
MEDTECH INC. |
| (Exact
name of registrant as specified in its charter) |
| Texas |
|
001-39678 |
|
59-2219994 |
| (State
or other jurisdiction of |
|
(Commission
File Number) |
|
(IRS
Employer |
| incorporation) |
|
|
|
Identification
No.) |
1200
Summit Avenue, Suite 414
Fort
Worth, Texas |
|
76102 |
| (Address
of principal executive offices) |
|
(Zip
Code) |
Registrant’s
telephone number, including area code: (817) 529-2300
(Former
name or former address, if changed since last report)
Not
Applicable
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under
any of the following provisions (see General Instruction A.2. below):
| |
☐ |
Written
communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| |
|
|
| |
☐ |
Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| |
|
|
| |
☐ |
Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| |
|
|
| |
☐ |
Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading
Symbol(s) |
|
Name
of each exchange on which registered |
| Common
Stock, $0.001 par value |
|
SMTI |
|
The
Nasdaq Capital Market |
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405
of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company ☐
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item
2.02 | Results
of Operations and Financial Condition. |
On
August 14, 2023, Sanara MedTech Inc. (the “Company”) issued a press release announcing its financial results for the quarter
ended June 30, 2023. A copy of the Company’s press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K and
is incorporated by reference herein.
In
addition, the Company is furnishing a copy of an earnings presentation (the “Presentation”) that the Company intends to use,
in whole or in part, in one or more meetings with investors or analysts, including in a webcast on August 15, 2023 at 9:00 a.m. (Eastern
Time). A copy of the Presentation is furnished as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated by reference herein.
The
information in this Current Report on Form 8-K, including Exhibits 99.1 and 99.2 furnished hereto, shall not be deemed “filed”
for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject
to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933,
as amended, or the Exchange Act, except as expressly set forth in such filing.
| Item
9.01 | Financial
Statements and Exhibits. |
(d)
Exhibits
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by
the undersigned hereunto duly authorized.
| Date: |
August
14, 2023 |
|
|
| |
|
|
|
| |
|
Sanara
MedTech Inc. |
| |
|
|
|
| |
|
By: |
/s/
Michael D. McNeil |
| |
|
Name: |
Michael D. McNeil |
| |
|
Title: |
Chief Financial Officer |
Exhibit
99.1

Sanara
MedTech Inc. Announces Second Quarter 2023 Results
FORT
WORTH, TX / GlobeNewswire / August 14, 2023 / Sanara MedTech Inc. Based in Fort Worth, Texas, Sanara MedTech Inc. (“Sanara,”
the “Company,” “we,” “our” or “us”) (NASDAQ: SMTI), a medical technology company focused
on developing and commercializing transformative technologies to improve clinical outcomes and reduce healthcare expenditures in the
surgical, chronic wound and skincare markets, announced today its strategic, operational and financial results for the quarter ended
June 30, 2023.
Zack
Fleming, Sanara’s CEO stated, “The second quarter of 2023 was another record revenue quarter for the Company. Subsequent
to the end of the quarter, we completed the acquisition of certain assets related to our collagen products business that we believe will
significantly add to our businesses’ performance by decreasing costs and eliminating the royalties we paid on CellerateRX Surgical
and HYCOL to the sellers. Additionally, we believe the acquisition will allow us to further develop and commercialize new efficacious
products.”
Second
Quarter 2023 Strategic and Operational Highlights (Unaudited)
| |
● |
During the trailing twelve-month period, the Company’s
products were sold in over 950 facilities across 33 states. |
| |
|
|
| |
● |
The Company’s products were contracted or approved to be sold in more than 3,000 hospitals/ASCs as of June 30, 2023. The large increase
quarter over quarter was attributable to the Company signing an agreement with a major group purchasing organization. |
| |
|
|
| |
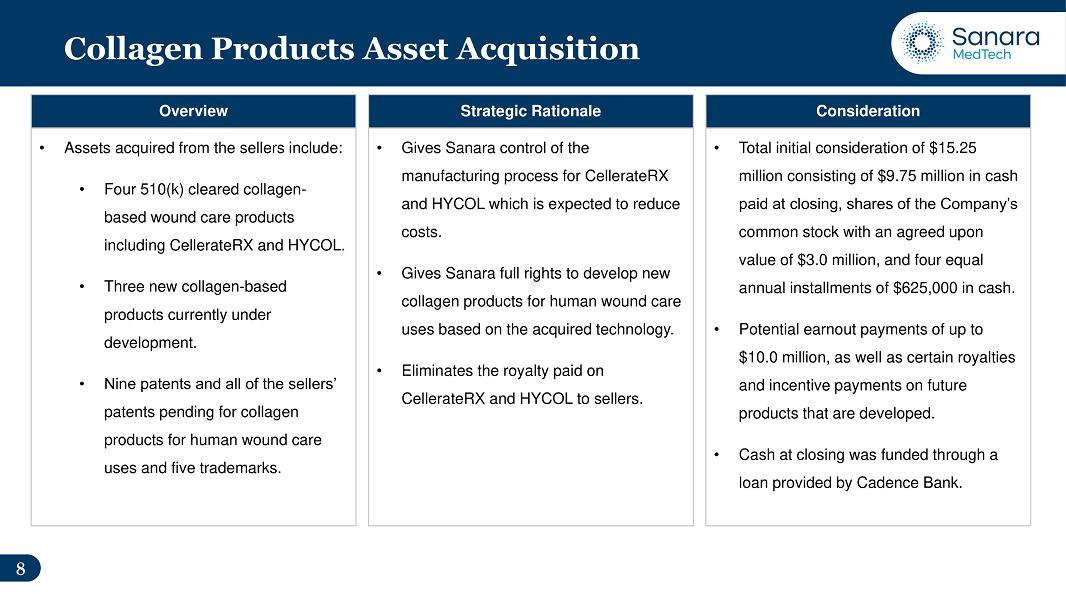
● |
Subsequent to the end of the quarter, the Company completed the acquisition of certain assets related to its collagen products business.
The assets acquired included, among others: |
| |
– |
All
rights and ownership (for human wound care uses) for four 510(k) cleared collagen-based wound care products, including CellerateRX®
Surgical Powder and Gel (“CellerateRX Surgical”) and HYCOL® Hydrolyzed Collagen (“HYCOL”). |
| |
|
|
| |
– |
All
rights and ownership (for human wound care uses) for three new collagen-based products currently under development. |
| |
|
|
| |
– |
All
patents, patents pending, trademarks and regulatory approvals related to collagen human wound care products owned by the sellers.
This includes nine patents and all of the sellers’ patents pending for collagen products for human wound care uses and five
trademarks. |
Sales
Analysis
The
Company generated net revenues of $15.8 million for the three months ended June 30, 2023, compared to net revenues of $9.7 million for
the three months ended June 30, 2022, a 63% increase from the prior year period. The higher net revenues for the three months ended June
30, 2023 were primarily due to increased sales of soft tissue repair products (CellerateRX Surgical, FORTIFY TRG® Tissue
Repair Graft, FORTIFY FLOWABLE® ECM, and TEXAGEN® Amniotic Membrane Allograft) and, to a lesser extent,
bone fusion products (BiFORM® Bioactive Moldable Matrix, AMPLIFY™ Verified Inductive Bone Matrix, and ALLOCYTE®
Advanced Cellular Bone Matrix), as a result of the Company’s increased market penetration and geographic expansion, additional
revenues as a result of the Company’s acquisition of Scendia Biologics, LLC in 2022 and the Company’s continuing strategy
to expand its independent distribution network in both new and existing U.S. markets. The Company’s sales continue to be negatively
impacted by supply issues related to its ALLOCYTE® Advanced Cellular Bone Matrix product. The Company is starting to see
improvement and expects an alternative source to come online in the near future.
Earnings
Analysis
The
Company had a loss before income taxes of $1.9 million for the three months ended June 30, 2023, compared to a loss before income taxes
of $3.4 million for the three months ended June 30, 2022. The lower loss before income taxes for the three months ended June 30, 2023
was due to operating expenses increasing at a slower rate than net sales in addition to the benefit recorded as a result of the change
in fair value of earnout liabilities. For the three months ended June 30, 2023, the Company had a net loss of $1.9 million, compared
to net income of $0.8 million for the three months ended June 30, 2022. The higher net income in 2022 was primarily due to a one-time
noncash income tax benefit realized in Q2 of 2022. Subsequent to the end of the quarter, the Company
completed the acquisition of certain assets related to its collagen products business. The Company believes that cost savings and the
elimination of royalties paid to the sellers will meaningfully contribute to the Company’s earnings going forward. In 2022, the
Company paid $1.8 million in royalties on its collagen products.
Conference
Call
Sanara
will host a conference call on Tuesday, August 15, 2023, at 9:00 a.m. Eastern Time. The toll-free number to call for this teleconference
is 888-506-0062 (international
callers: 973-528-0011) and the access code is 233776. A telephonic replay of the conference call will be available through Tuesday, August
29, 2023, by dialing 877-481-4010 (international callers: 919-882-2331) and entering the replay passcode: 48852.
A
live webcast of Sanara’s conference call will be available under the Investor Relations section of the Company’s website,
www.SanaraMedTech.com. A one-year online replay will be available after the conclusion of the live broadcast.
About
Sanara MedTech Inc.
With
a focus on improving patient outcomes through evidence-based healing solutions, Sanara MedTech Inc. markets, distributes and develops
surgical, wound and skincare products for use by physicians and clinicians in hospitals, clinics and all post-acute care settings and
offers wound care and dermatology virtual consultation services via telemedicine. Sanara’s products are primarily sold in the North
American advanced wound care and surgical tissue repair markets. Sanara markets and distributes CellerateRX® Surgical Activated Collagen®,
FORTIFY TRG® Tissue Repair Graft and FORTIFY FLOWABLE® Extracellular Matrix as well as a portfolio of advanced biologic products
focusing on AMPLIFY™ Verified Inductive Bone Matrix, ALLOCYTE® Advanced Cellular Bone Matrix, BiFORM® Bioactive Moldable
Matrix and TEXAGEN® Amniotic Membrane Allograft to the surgical market. In addition, the following products are sold in the wound
care market: BIAKŌS® Antimicrobial Skin and Wound Cleanser, BIAKŌS™ Antimicrobial Wound Gel, BIAKŌS®
Antimicrobial Skin and Wound Irrigation Solution and HYCOL® Hydrolyzed Collagen. Sanara’s pipeline also contains potentially
transformative product candidates for mitigation of opportunistic pathogens and biofilm, wound re-epithelialization and closure, necrotic
tissue debridement and cell compatible substrates. The Company believes it has the ability to drive its pipeline from concept to preclinical
and clinical development while meeting quality and regulatory requirements. Sanara is constantly seeking long-term strategic partnerships
with a focus on products that improve outcomes at a lower overall cost. In addition, Sanara is actively seeking to expand within its
six focus areas of wound and skin care for the acute, post-acute, and surgical markets. The focus areas are debridement, biofilm removal,
hydrolyzed collagen, advanced biologics, negative pressure wound therapy products and the oxygen delivery system segment of the wound
and skincare markets.
Information
about Forward-Looking Statements
The
statements in this press release that do not constitute historical facts are “forward-looking statements,” within the meaning
of and subject to the safe harbor created by the Private Securities Litigation Reform Act of 1995. These statements may be identified
by terms such as “aims,” “anticipates,” “believes,” “contemplates,” “continue,”
“could,” “estimates,” “expect,” “forecast,” “guidance,” “intend,”
“may,” “plan,” “possible,” “potential,” “predicts,” “preliminary,”
“projects,” “seeks,” “should,” “targets,” “will” or “would,”
or the negatives of these terms, variations of these terms or other similar expressions. These forward-looking statements include, among
others, statements regarding the potential benefits created by the acquisition of certain assets related to the Company’s collagen
products business, the anticipated impact of such acquisition on the Company’s business and future financial and operating results,
the Company’s ability to develop and commercialize the new collagen-based products currently under development, including the manufacturing,
distribution, marketing and sale of such products, the Company’s ability to maintain or replace the manufacturing and distribution
process of the sellers in the acquisition, including relationships with vendors, the development of new products, the timing of commercialization
of our products, the regulatory approval process and expansion of the Company’s business in telehealth and wound care. These items
involve risks, contingencies and uncertainties such as the extent of product demand, market and customer acceptance, the effect of economic
conditions, competition, pricing, uncertainties associated with the development and process for obtaining regulatory approval for new
products, the ability to consummate and integrate acquisitions, and other risks, contingencies and uncertainties detailed in the Company’s
SEC filings, which could cause the Company’s actual operating results, performance or business plans or prospects to differ materially
from those expressed in, or implied by these statements.
All
forward-looking statements speak only as of the date on which they are made, and the Company undertakes no obligation to revise any of
these statements to reflect the future circumstances or the occurrence of unanticipated events, except as required by applicable securities
laws.
Investor
Contact:
Callon
Nichols, Director of Investor Relations
713-826-0524
CNichols@sanaramedtech.com
SOURCE:
Sanara MedTech Inc.
SANARA
MEDTECH INC. AND SUBSIDIARIES
CONSOLIDATED
BALANCE SHEETS
| | |
(Unaudited) | | |
| |
| | |
June 30, | | |
December 31, | |
| | |
2023 | | |
2022 | |
| Assets | |
| | | |
| | |
| Current assets | |
| | | |
| | |
| Cash | |
$ | 6,060,228 | | |
$ | 8,958,995 | |
| Accounts receivable, net | |
| 7,141,105 | | |
| 6,805,761 | |
| Accounts receivable – related party | |
| 20,662 | | |
| 98,548 | |
| Royalty receivable | |
| 49,344 | | |
| 99,594 | |
| Inventory, net | |
| 4,420,864 | | |
| 3,549,000 | |
| Prepaid and other assets | |
| 485,734 | | |
| 1,104,611 | |
| Total current assets | |
| 18,177,937 | | |
| 20,616,509 | |
| | |
| | | |
| | |
| Long-term assets | |
| | | |
| | |
| Property and equipment, net | |
| 1,240,269 | | |
| 1,416,436 | |
| Right of use assets – operating leases | |
| 2,030,938 | | |
| 806,402 | |
| Goodwill | |
| 3,601,781 | | |
| 3,601,781 | |
| Intangible assets, net | |
| 30,143,578 | | |
| 31,509,980 | |
| Investment in equity securities | |
| 3,084,278 | | |
| 3,084,278 | |
| Total long-term assets | |
| 40,100,844 | | |
| 40,418,877 | |
| | |
| | | |
| | |
| Total assets | |
$ | 58,278,781 | | |
$ | 61,035,386 | |
| | |
| | | |
| | |
| Liabilities and shareholders’ equity | |
| | | |
| | |
| Current liabilities | |
| | | |
| | |
| Accounts payable | |
$ | 1,016,180 | | |
$ | 1,392,701 | |
| Accounts payable – related parties | |
| 96,656 | | |
| 34,036 | |
| Accrued royalties and expenses | |
| 1,895,706 | | |
| 2,144,475 | |
| Accrued bonuses and commissions | |
| 5,899,255 | | |
| 7,758,284 | |
| Earnout liabilities – current | |
| 700,000 | | |
| 1,162,880 | |
| Operating lease liabilities – current | |
| 273,539 | | |
| 313,933 | |
| Total current liabilities | |
| 9,881,336 | | |
| 12,806,309 | |
| | |
| | | |
| | |
| Long-term liabilities | |
| | | |
| | |
| Earnout liabilities – long-term | |
| 5,653,534 | | |
| 6,003,811 | |
| Operating lease liabilities – long-term | |
| 1,779,413 | | |
| 505,291 | |
| Total long-term liabilities | |
| 7,432,947 | | |
| 6,509,102 | |
| | |
| | | |
| | |
| Total liabilities | |
| 17,314,283 | | |
| 19,315,411 | |
| | |
| | | |
| | |
| Commitments and contingencies | |
| | | |
| | |
| | |
| | | |
| | |
| Shareholders’ equity | |
| | | |
| | |
| Common Stock: $0.001 par value, 20,000,000 shares authorized; 8,439,745 issued and outstanding as of
June 30, 2023 and 8,299,957 issued and outstanding as of December 31, 2022 | |
| 8,440 | | |
| 8,300 | |
| Additional paid-in capital | |
| 67,881,419 | | |
| 65,213,987 | |
| Accumulated deficit | |
| (26,740,930 | ) | |
| (23,394,757 | ) |
| Total Sanara MedTech shareholders’ equity | |
| 41,148,929 | | |
| 41,827,530 | |
| Equity attributable to noncontrolling interest | |
| (184,431 | ) | |
| (107,555 | ) |
| Total shareholders’ equity | |
| 40,964,498 | | |
| 41,719,975 | |
| Total liabilities and shareholders’ equity | |
$ | 58,278,781 | | |
$ | 61,035,386 | |
SANARA
MEDTECH INC. AND SUBSIDIARIES
CONSOLIDATED
STATEMENTS OF OPERATIONS (UNAUDITED)
| | |
Three Months Ended | | |
Six Months Ended | |
| | |
June 30, | | |
June 30, | |
| | |
2023 | | |
2022 | | |
2023 | | |
2022 | |
| | |
| | |
| | |
| | |
| |
| Net Revenue | |
$ | 15,753,164 | | |
$ | 9,670,778 | | |
$ | 31,275,081 | | |
$ | 17,482,001 | |
| | |
| | | |
| | | |
| | | |
| | |
| Cost of goods sold | |
| 2,187,516 | | |
| 958,086 | | |
| 4,313,175 | | |
| 1,763,167 | |
| | |
| | | |
| | | |
| | | |
| | |
| Gross profit | |
| 13,565,648 | | |
| 8,712,692 | | |
| 26,961,906 | | |
| 15,718,834 | |
| | |
| | | |
| | | |
| | | |
| | |
| Operating expenses | |
| | | |
| | | |
| | | |
| | |
| Selling, general and administrative expenses | |
| 13,811,476 | | |
| 10,428,133 | | |
| 26,780,545 | | |
| 19,803,763 | |
| Research and development | |
| 1,177,128 | | |
| 1,067,000 | | |
| 2,494,452 | | |
| 1,271,637 | |
| Depreciation and amortization | |
| 803,694 | | |
| 539,124 | | |
| 1,582,569 | | |
| 741,871 | |
| Change in fair value of earnout liabilities | |
| (360,470 | ) | |
| 63,427 | | |
| (813,157 | ) | |
| 63,427 | |
| Total operating expenses | |
| 15,431,828 | | |
| 12,097,684 | | |
| 30,044,409 | | |
| 21,880,698 | |
| | |
| | | |
| | | |
| | | |
| | |
| Operating loss | |
| (1,866,180 | ) | |
| (3,384,992 | ) | |
| (3,082,503 | ) | |
| (6,161,864 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Other expense | |
| | | |
| | | |
| | | |
| | |
| Interest expense and other | |
| - | | |
| - | | |
| (6 | ) | |
| - | |
| Share of losses from equity method investment | |
| - | | |
| - | | |
| - | | |
| (379,633 | ) |
| Total other expense | |
| - | | |
| - | | |
| (6 | ) | |
| (379,633 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Loss before income taxes | |
| (1,866,180 | ) | |
| (3,384,992 | ) | |
| (3,082,509 | ) | |
| (6,541,497 | ) |
| Income tax benefit | |
| - | | |
| 4,141,906 | | |
| - | | |
| 4,141,906 | |
| | |
| | | |
| | | |
| | | |
| | |
| Net income (loss) | |
| (1,866,180 | ) | |
| 756,914 | | |
| (3,082,509 | ) | |
| (2,399,591 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Less: Net loss attributable to noncontrolling interest | |
| (38,447 | ) | |
| (12,512 | ) | |
| (76,876 | ) | |
| | |
| | |
| | | |
| | | |
| | | |
| (39,693 | ) |
| Net income (loss) attributable to Sanara MedTech
shareholders | |
$ | (1,827,733 | ) | |
$ | 769,426 | | |
$ | (3,005,633 | ) | |
$ | (2,359,898 | ) |
| | |
| | | |
| | | |
| | | |
| | |
| Net income (loss) per share: | |
| | | |
| | | |
| | | |
| | |
| Basic | |
$ | (0.22 | ) | |
$ | 0.10 | | |
$ | (0.37 | ) | |
$ | (0.31 | ) |
| Diluted | |
$ | (0.22 | ) | |
$ | 0.09 | | |
$ | (0.37 | ) | |
$ | (0.31 | ) |
| Weighted average common shares outstanding: | |
| | | |
| | | |
| | | |
| | |
| Basic | |
| 8,226,271 | | |
| 7,791,431 | | |
| 8,200,173 | | |
| 7,699,422 | |
| Diluted | |
| 8,226,271 | | |
| 8,162,983 | | |
| 8,200,173 | | |
| 7,699,422 | |
SANARA
MEDTECH INC. AND SUBSIDIARIES
CONSOLIDATED
STATEMENTS OF CASH FLOWS (UNAUDITED)
| | |
Six Months Ended | |
| | |
June 30, | |
| | |
2023 | | |
2022 | |
| | |
| | |
| |
| Cash flows from operating activities: | |
| | | |
| | |
| Net loss | |
$ | (3,082,509 | ) | |
$ | (2,399,591 | ) |
| Adjustments to reconcile net loss to net cash used in operating activities: | |
| | | |
| | |
| Depreciation and amortization | |
| 1,582,569 | | |
| 741,871 | |
| Loss on disposal of property and equipment | |
| - | | |
| 2,500 | |
| Bad debt expense | |
| 86,000 | | |
| 195,000 | |
| Inventory obsolescence | |
| 69,990 | | |
| 159,717 | |
| Share-based compensation | |
| 1,724,637 | | |
| 1,288,335 | |
| Noncash lease expense | |
| 144,628 | | |
| 116,143 | |
| Loss on equity method investment | |
| - | | |
| 379,633 | |
| Benefit from deferred income taxes | |
| - | | |
| (4,141,906 | ) |
| Change in fair value of earnout liabilities | |
| (813,157 | ) | |
| 63,427 | |
| Changes in operating assets and liabilities: | |
| | | |
| | |
| Accounts receivable, net | |
| (371,094 | ) | |
| (1,170,829 | ) |
| Accounts receivable – related party | |
| 77,886 | | |
| (130,797 | ) |
| Inventory, net | |
| (941,854 | ) | |
| (423,764 | ) |
| Prepaid and other assets | |
| 618,877 | | |
| 177,861 | |
| Accounts payable | |
| (376,521 | ) | |
| 294,772 | |
| Accounts payable – related parties | |
| 62,620 | | |
| 589,675 | |
| Accrued royalties and expenses | |
| (248,769 | ) | |
| 761,377 | |
| Accrued bonuses and commissions | |
| (1,859,029 | ) | |
| 346,887 | |
| Operating lease liabilities | |
| (135,436 | ) | |
| (117,106 | ) |
| Net cash used in operating activities | |
| (3,461,162 | ) | |
| (3,266,795 | ) |
| Cash flows from investing activities: | |
| | | |
| | |
| Purchases of property and equipment | |
| (40,650 | ) | |
| (80,892 | ) |
| Proceeds from disposal of assets | |
| 650 | | |
| 345 | |
| Purchases of intangible assets | |
| - | | |
| (2,053,722 | ) |
| Investment in equity securities | |
| - | | |
| (250,000 | ) |
| Net cash used in investing activities | |
| (40,000 | ) | |
| (2,384,269 | ) |
| Cash flows from financing activities: | |
| | | |
| | |
| Equity offering net proceeds | |
| 1,033,761 | | |
| - | |
| Net settlement of equity-based awards | |
| (431,366 | ) | |
| (102,931 | ) |
| Distribution to noncontrolling interest member | |
| - | | |
| (220,000 | ) |
| Net cash provided by (used in) financing activities | |
| 602,395 | | |
| (322,931 | ) |
| Net decrease in cash | |
| (2,898,767 | ) | |
| (5,973,995 | ) |
| Cash, beginning of period | |
| 8,958,995 | | |
| 18,652,841 | |
| Cash, end of period | |
$ | 6,060,228 | | |
$ | 12,678,846 | |
| | |
| | | |
| | |
| Cash paid during the period for: | |
| | | |
| | |
| Interest | |
$ | 6 | | |
$ | - | |
| Supplemental noncash investing and financing activities: | |
| | | |
| | |
| Right of use assets obtained in exchange for lease obligations | |
| 1,369,164 | | |
| - | |
| Equity issued for acquisitions | |
| - | | |
| 9,709,089 | |
| Earnout liabilities generated by acquisitions | |
| - | | |
| 3,882,151 | |
| Investment in equity securities converted in asset acquisition | |
| - | | |
| 1,803,440 | |
Exhibit 99.2










v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Sanara MedTech (NASDAQ:SMTI)
Graphique Historique de l'Action
De Mai 2024 à Juin 2024

Sanara MedTech (NASDAQ:SMTI)
Graphique Historique de l'Action
De Juin 2023 à Juin 2024
