Insight Pharmaceuticals Issues Voluntary Nationwide Recall of Americaine® 20% Benzocaine Topical Anesthetic Spray Due to the Presence of Benzene
22 Décembre 2023 - 3:00PM

Insight Pharmaceuticals, a Prestige Consumer Healthcare Inc.
company (“Insight”), is voluntarily recalling one lot of
Americaine® 20% Benzocaine Topical Anesthetic Spray to the consumer
level. While benzene is not an ingredient in any Americaine® 20%
Benzocaine Topical Anesthetic Spray products, a recent review by
our manufacturer and their third-party lab found that a sample from
one lot of the product showed a low level of benzene came from the
propellant that sprays the product out of the can. Importantly, no
other lots of Americaine® 20% Benzocaine Topical Anesthetic Spray
(either before or after this batch code) are in the scope of this
recall and may continue to be used by consumers safely and as
intended.
Risk Statement: Benzene is classified as a human
carcinogen. Exposure to benzene can occur by inhalation, orally,
and through the skin and it potentially can result in cancers
including leukemia and blood cancer of the bone marrow and blood
disorders which can be life threatening. Insight is recalling these
products out of an abundance of caution. To date, the Company has
not received any serious adverse events related to this recall.
Benzene is ubiquitous in the environment. Humans around the world
have daily exposures to it indoors and outdoors from multiple
sources.
Americaine® 20% Benzocaine Topical Anesthetic
Spray is packaged in white aerosol cans with Teal caps with Lot
codes located on the bottom of the can.
Samples of the recalled lot below have been found
to contain trace levels of Benzene related to the propellant that
sprays the product out of the can. Benzene is not an ingredient in
any Americaine products.
|
Product |
NDC |
Lot Code |
Expiration |
Package Size |
|
Americaine® 20% Benzocaine Topical Anesthetic Spray |
63736-378-02 |
1A16420 |
01/25 |
2 oz / 57 g |
The affected Americaine® 20% Benzocaine Topical
Anesthetic Spray lot was distributed nationwide in the United
States through a limited number of retailers and online.
Insight has notified retailers via overnight mail
to remove any remaining recalled product from shelves and follow
the instructions provided in the Drug Recall Notification. The
company will also offer reimbursement for consumers who have
purchased Americaine® 20% Benzocaine Topical Anesthetic Spray
marked with the lot code in the table above. Consumers can contact
Insight Pharmaceuticals via e-mail at
medicalaffairs@prestigebrands.com, through its website at
https://www.prestigebrands.com/contact, or by phone at (800)
344-7239 on Monday – Friday 8:30-5:30 eastern time to receive a
full refund by providing a picture of the bottom of the can of the
Americaine® 20% Benzocaine Topical Anesthetic Spray with the
affected lot number. Consumers that have product which is being
recalled should stop using the product immediately
and appropriately discard after taking the picture. Consumers
should contact their physician or healthcare provider if they have
experienced any problems that may be related to taking or using
this anesthetic product.
No serious adverse events have been reported to
date.
Adverse reactions or quality problems experienced
with the use of this product may be reported to the FDA's MedWatch
Adverse Event Reporting program either online, by regular mail or
by fax.
- Complete and submit the report Online:
www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form
www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request
a reporting form, then complete and return to the address on the
pre-addressed form, or submit by fax to 1-800-FDA-0178.
This recall is being conducted with the knowledge
of the U.S. Food and Drug Administration.
A photo accompanying this announcement is available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/b32d23b6-9edf-47e7-9c8d-2f763b65b560.
Investor Relations Contact
Phil Terpolilli, CFA, 914-524-6819
irinquiries@prestigebrands.com
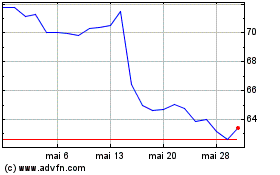
Prestige Consumer Health... (NYSE:PBH)
Graphique Historique de l'Action
De Avr 2024 à Mai 2024
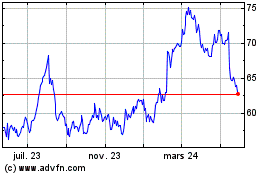
Prestige Consumer Health... (NYSE:PBH)
Graphique Historique de l'Action
De Mai 2023 à Mai 2024