Johnson & Johnson to Begin Human Trials on Covid-19 Vaccine by Sept
30 Mars 2020 - 2:09PM
Dow Jones News
By Matt Grossman
Johnson & Johnson said Monday that it has made progress on a
vaccine to prevent Covid-19 and the product could be ready in early
2021.
The New Brunswick, N.J.-based company selected a lead candidate
for the vaccine from the constructs it had been working on, and
said human trials will begin by September at the latest. It said it
could get approval under an emergency-use authorization to make the
vaccine widely available early next year.
Johnson & Johnson said it would try to scale its global
manufacturing capabilities to make more than 1 billion of a
Covid-19 vaccine. It said it is also working with the Department of
Health and Human Services to expand its research into a treatment
for Covid-19.
Write to Matt Grossman at matt.grossman@wsj.com
(END) Dow Jones Newswires
March 30, 2020 07:54 ET (11:54 GMT)
Copyright (c) 2020 Dow Jones & Company, Inc.
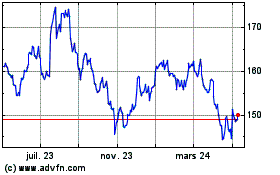
Johnson and Johnson (NYSE:JNJ)
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
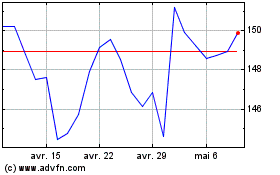
Johnson and Johnson (NYSE:JNJ)
Graphique Historique de l'Action
De Avr 2023 à Avr 2024