- This new treatment provides adults living with schizophrenia
a long-acting formulation that offers flexible 1- and 2-month
dosing intervals1
- In a Phase 3 clinical trial, UZEDY demonstrated up to 80%
reduction in risk of schizophrenia relapse versus placebo1
- UZEDY is a subcutaneous injection from a pre-filled syringe
with a 21-gauge needle
Teva Pharmaceuticals, a U.S. affiliate of Teva Pharmaceutical
Industries Ltd. (NYSE and TASE: TEVA), and MedinCell (Euronext:
MEDCL) announced today that the U.S. Food and Drug Administration
(FDA) has approved UZEDY (risperidone) extended-release injectable
suspension for the treatment of schizophrenia in adults. UZEDY is
the first subcutaneous, long-acting formulation of risperidone that
utilizes SteadyTeq™, a copolymer technology proprietary to
MedinCell that controls the steady release of risperidone.
Therapeutic blood concentrations are reached within 6-24 hours of a
single dose.1
“UZEDY embodies Teva’s commitment to bringing innovative
advances to patients and to providing people living with
schizophrenia an important new treatment option that was designed
to address certain treatment challenges and may decrease the risk
of relapse,” said Richard Francis, President and CEO of Teva. “The
approval of UZEDY is a culmination of a multidisciplinary effort
across Teva and MedinCell to bring this important treatment to
market. This milestone is a testament to advancing our robust
biopharmaceutical pipeline of innovative medicines that aim to
support more people living with mental health disorders and
neurological diseases in the coming years.”
Approximately 80% of patients with schizophrenia experience
multiple relapses over the first five years of treatment,2 most
commonly due to suboptimal adherence to treatment with oral
antipsychotics. Each relapse carries a biological risk of loss of
function, treatment refractoriness, and changes in brain
morphology.3,4
Schizophrenia is a chronic, progressive and severely
debilitating mental health disorder that affects how one thinks,
feels and acts.5 This approval is based on data from two Phase 3
trials evaluating UZEDY in patients with schizophrenia:
TV46000-CNS-30072 (the RISE Study – The Risperidone Subcutaneous
Extended-Release Study) and TV46000-CNS-30078 (the SHINE Study – A
Study to Test TV-46000 for Maintenance Treatment of
Schizophrenia).
"The approval of the first product formulated with our
technology is a pivotal moment for MedinCell and for the many
patients who will benefit,” said Christophe Douat, CEO of
MedinCell. “We are committed to supporting patients through
innovative therapy options. It continues to be a wonderful journey
with Teva, an ideal partner to harness the full potential of UZEDY.
Our technology reaching commercial stage marks the start of an
exciting new era for MedinCell and we are extremely proud to share
this very special moment with all our employees and
shareholders."
The use of novel SteadyTeq technology in UZEDY controls the
release of risperidone over time. The initiation of treatment
requires no loading dose or oral supplementation. Therapeutic blood
concentrations are reached within 6-24 hours of a single dose.1
“Treatments for schizophrenia are largely prescribed as daily
oral medications, which can present challenges with adherence due
to missed doses. Lack of adherence to treatment with oral
antipsychotics is the most common cause of relapse in
schizophrenia,6 so there’s a role for therapies that are dosed in
one- or two-month dosing intervals to help prevent relapse,” said
Christoph Correll, MD, professor of psychiatry at the Zucker School
of Medicine, Hempstead, NY. “As a clinician, I am excited to now
have a new treatment option that reduces the risk of relapse1 for
this complex disease and helps address some of the barriers around
receiving schizophrenia treatment.”
CLICK HERE TO ACCESS THE COMPLETE PRESS RELEASE
About Schizophrenia
Schizophrenia is a chronic, progressive and severely
debilitating mental disorder that affects how one thinks, feels and
acts. Patients experience an array of symptoms, which may include
delusions, hallucinations, disorganized speech or behavior and
impaired cognitive ability. Approximately 1% of the world’s
population will develop schizophrenia in their lifetime, and 3.5
million people in the U.S. are currently diagnosed with the
condition. Although schizophrenia can occur at any age, the average
age of onset tends to be in the late teens to the early 20s for
men, and the late 20s to early 30s for women. The long-term course
of schizophrenia is marked by episodes of partial or full remission
broken by relapses that often occur in the context of psychiatric
emergency and require hospitalization. Approximately 80% of
patients experience multiple relapses over the first five years of
treatment, and each relapse carries a biological risk of loss of
function, treatment refractoriness, and changes in brain
morphology. Patients are often unaware of their illness and its
consequences, contributing to treatment nonadherence, high
discontinuation rates, and ultimately, significant direct and
indirect healthcare costs from subsequent relapses and
hospitalizations.
About UZEDY
UZEDY (risperidone) extended-release injectable suspension, for
subcutaneous use rather than intramuscular use, is indicated for
the treatment of schizophrenia in adults. In clinical trials, UZEDY
reduced the risk of relapse by up to 80%. UZEDY administers
risperidone through copolymer technology under license from
MedinCell that allows for absorption and sustained release in the
first subcutaneous injection. UZEDY is the only long-acting,
subcutaneous formulation of risperidone available in both one- and
two-month dosing intervals.1 For full prescribing information,
visit
https://www.uzedy.com/globalassets/uzedy/prescribing-information.pdf.
About Teva
Teva Pharmaceutical Industries Ltd. (NYSE and TASE: TEVA) has
been developing and producing medicines to improve people’s lives
for more than a century. We are a global leader in generic,
biosimilar and innovative medicines with a portfolio consisting of
over 3,500 products in nearly every therapeutic area. Around 200
million people around the world take a Teva medicine every day, and
are served by one of the largest and most complex supply chains in
the pharmaceutical industry. Along with our established presence in
generics, we have significant innovative research and operations
supporting our growing portfolio of innovative medicines and
biopharmaceutical products. Learn more at www.tevapharm.com.
About MedinCell
MedinCell is an innovative pharmaceutical company with a
portfolio of long-acting injectable products, from development to
market, in various therapeutic areas. MedinCell proprietary
technology BEPO® (licensed to Teva under the name SteadyTeq) makes
it possible to control the delivery of a drug at a therapeutic dose
for several days, weeks or months starting from the subcutaneous or
local injection of a simple deposit of a few millimeters, fully
bioresorbable. MedinCell collaborate with tier one pharmaceuticals
companies and foundations to improve Global Health through new
therapeutic options. Based in Montpellier, MedinCell currently
employs more than 140 people representing over 25 different
nationalities. www.medincell.com
1 UZEDY™ (risperidone) extended-release injectable suspension,
for subcutaneous injection Current Prescribing Information.
Parsippany, NJ. Teva Neuroscience, Inc.
2 Emsley, R., & Kilian, S. (2018). Efficacy and safety
profile of paliperidone palmitate injections in the management of
patients with schizophrenia: an evidence-based review.
Neuropsychiatric disease and treatment, 14, 205–223.
3 Emsley, R., Chiliza, B., Asmal, L. et al. (2013) The nature of
relapse in schizophrenia. BMC Psychiatry 13, 50.
4 Andreasen, N. C., et al. (2013). Relapse duration, treatment
intensity, and brain tissue loss in schizophrenia: a prospective
longitudinal MRI study. The American journal of psychiatry, 170(6),
609–615.
5 Patel, K. R., Cherian, J., Gohil, K., & Atkinson, D.
(2014). Schizophrenia: overview and treatment options. P & T: a
peer-reviewed journal for formulary management, 39(9),
638–645..
6 Kane JM, Correll CU. Optimizing treatment choices to improve
adherence and outcomes in schizophrenia. J Clin Psychiatry.
2019;80(5):IN18031AH1C. doi:10.4088/JCP.IN18031AH1C.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20230428005632/en/
IR Contacts Ran Meir, +1 (267) 468-4475 Yael Ashman, +972
(3) 914 8262 Sanjeev Sharma, +1 (973) 658 2700
PR Contacts Kelley Dougherty, 1 (973) 832-2810 Eden
Klein, +972 (3) 906 2645
MedinCell France David Heuzé (Head of Communication), +33
(0) 6 83 25 21 86 david.heuze@medincell.com
Louis-Victor Delouvrier (NewCap – IR), +33 (0) 1 44 71 98 53
medincell@newcap.eu
Nicolas Merigeau (NewCap - Media Relations), +33 (0) 1 44 71 94
98 medincell@newcap.eu
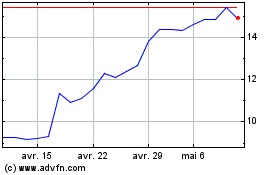
Medincell (EU:MEDCL)
Graphique Historique de l'Action
De Avr 2024 à Mai 2024
Medincell (EU:MEDCL)
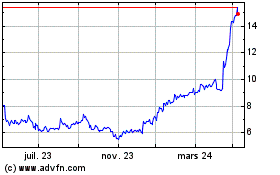
Graphique Historique de l'Action
De Mai 2023 à Mai 2024