Teva, MedinCell Say Results Positive in Risperidone Trial
07 Janvier 2021 - 3:12PM
Dow Jones News
By Michael Dabaie
Teva Pharmaceuticals Industries Ltd. and MedinCell on Thursday
said results were positive for Phase 3 clinical trial of
TV-46000/mdc-IRM as a treatment for patients with
schizophrenia.
Trial enrollment was open to patients 13-65 years of age. In the
RISE study, patients treated with the investigational subcutaneous
risperidone injection either monthly or once every two months
experienced a statistically significant delay in time to relapse
versus placebo, the study's primary objective.
No new safety signals have been identified that are inconsistent
with the known safety profile of other risperidone
formulations.
Teva will continue to lead the clinical development and
regulatory process and be responsible for commercialization of this
candidate treatment, with MedinCell eligible for development
milestones, royalties on net sales and future commercial
milestones.
Teva shares were up 0.7%, to $11.17, premarket.
Write to Michael Dabaie at michael.dabaie@wsj.com
(END) Dow Jones Newswires
January 07, 2021 08:57 ET (13:57 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
Medincell (EU:MEDCL)
Graphique Historique de l'Action
De Avr 2024 à Mai 2024
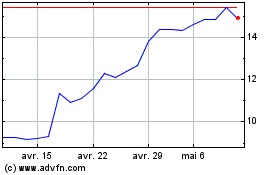
Medincell (EU:MEDCL)
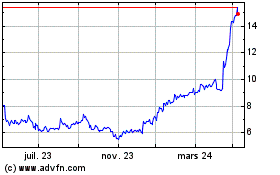
Graphique Historique de l'Action
De Mai 2023 à Mai 2024