AN2 Therapeutics to Present New Data for Epetraborole at IDWeek 2023
11 Octobre 2023 - 6:01AM
Business Wire
Enrollment Ongoing in Phase 3 Part of 2/3
Pivotal Trial of Epetraborole in Treatment-Refractory Mycobacterium
avium complex (MAC) Lung Disease
AN2 Therapeutics, Inc. (Nasdaq: ANTX), a clinical-stage
biopharmaceutical company focused on developing treatments for
rare, chronic, and serious infectious diseases with high unmet
needs, today announced two oral presentations and three poster
presentations at the Infectious Disease Society of America (IDSA)
IDWeek 2023 Conference from October 11-15, 2023 in Boston,
Massachusetts. AN2 Therapeutics is currently enrolling patients in
the Phase 3 part of its pivotal Phase 2/3 clinical trial evaluating
once-daily, oral epetraborole for treatment-refractory
Mycobacterium avium complex (MAC) lung disease.
“We are excited to showcase our latest data at IDWeek 2023.
These results highlight epetraborole’s potent nonclinical activity
against MAC isolates, as well as M. abscessus, another
difficult-to-treat NTM pathogen. Additionally, epetraborole
demonstrated favorable pharmacokinetics at the current dose in
patients with various degrees of renal impairment and in Japanese
subjects with different alcohol dehydrogenase genotypes, which
further supports our dosing strategy in patients with
treatment-refractory MAC lung disease,” said Paul Eckburg, M.D.,
Chief Medical Officer of AN2 Therapeutics. “These data, combined
with results from our ongoing pivotal Phase 2/3 study — which is
currently enrolling participants in the Phase 3 portion — are
expected to provide the basis for regulatory filings for approval
in the U.S. and Japan. We are enthusiastic about the progress we
have achieved so far and look forward to advancing our mission to
develop a novel therapy for patients with treatment-refractory MAC
lung disease."
Oral presentations pertaining to epetraborole:
Title: Epetraborole: A Novel Antibiotic for NTM Lung Disease
& Melioidosis Session: New Antimicrobials in the Pipeline Part
1 Date: Thursday, October 12, 2023 Time: 8:48 AM – 9:00 AM ET Room:
104ABC Presentation # 32
Title: In Vitro Susceptibility of Recent Mycobacterium abscessus
Isolates to Epetraborole (EBO) and Comparators by Broth
Microdilution Session: Experimental Therapeutics Date: Saturday,
October 14, 2023 Time: 10:54 AM – 11:06 AM ET Location: 102AB
Presentation # 2064
Poster presentations pertaining to epetraborole:
Title: A Phase 1, Multicenter, Open-Label, Parallel-Group Study
to Assess the Safety and Pharmacokinetics (PK) of Oral Epetraborole
Tablets in Adult Subjects with Varying Degrees of Renal Function
Session: Antimicrobial Novel Agents Date: Saturday, October 14,
2023 Time: 12:15 PM - 1:30 PM ET Location: Hall B&C
Presentation # 2144
Title: A Phase 1, Open-Label, Single Dose Study to Evaluate the
Pharmacokinetics (PK), Safety, and Tolerability of Epetraborole
Tablets and the Impact of Alcohol Dehydrogenase (ADH) Genotype on
the PK of Epetraborole and Metabolite M3 in Healthy Japanese Adult
Subjects Session: PK/PD Studies Date: Saturday, October 14, 2023
Time: 12:15 PM - 1:30 PM ET Location: Hall B&C Presentation #
2556
Title: Epetraborole in vitro Activity Against Mycobacterium
avium complex Recent Clinical Isolates from Japan Session:
Antimicrobial Novel Agents Date: Saturday, October 14, 2023 Time:
12:15 PM - 1:30 PM ET Location: Hall B&C Presentation #
2135
About AN2 Therapeutics, Inc. AN2 Therapeutics, Inc. is a
clinical-stage biopharmaceutical company developing treatments for
rare, chronic, and serious infectious diseases with high unmet
needs. Our initial candidate is epetraborole, which we are studying
as a once-daily, oral treatment with a novel mechanism of action
for patients with nontuberculous mycobacteria (NTM) lung disease, a
rare, chronic, and progressive infectious disease caused by
bacteria known as mycobacteria, that leads to irreversible lung
damage and can be fatal. For more information, please visit our
website at www.an2therapeutics.com.
Forward-Looking Statements This press release contains
forward-looking statements within the meaning of the Private
Securities Litigation Reform Act of 1995. Forward-looking
statements expressed or implied in this press release include, but
are not limited to, statements regarding: the timing, progress, and
anticipated results from AN2's Phase 2/3 pivotal clinical trial;
AN2's anticipated progress, business plans, business strategy and
planned clinical trials; the potential clinical benefits and
therapeutic potential of epetraborole; and other statements that
are not historical fact. These statements are based on AN2's
current estimates, expectations, plans, objectives, and intentions,
are not guarantees of future performance and inherently involve
significant risks and uncertainties. Actual results and the timing
of events could differ materially from those anticipated in such
forward-looking statements as a result of these risks and
uncertainties, which include, but are not limited to, risks and
uncertainties related to: the continuing effects of the COVID-19
pandemic; macroeconomic conditions; AN2's ability to progress
enrollment in its Phase 2/3 pivotal clinical trial of epetraborole;
the ability of AN2 to effectively and timely make amendments to the
Phase 2/3 pivotal trial design pursuant to additional FDA feedback;
timely enrollment of patients in its current and future clinical
trials; AN2's ability to procure sufficient supply of its product
candidate for its existing and future clinical trials; the
potential for results from clinical trials to differ from
preclinical, early clinical, preliminary or expected results;
significant adverse events, toxicities or other undesirable side
effects associated with AN2's product candidate; the significant
uncertainty associated with AN2's product candidate ever receiving
any regulatory approvals; AN2's ability to obtain, maintain or
protect intellectual property rights related to its current and
future product candidates; implementation of AN2's strategic plans
for its business and current and future product candidates; the
sufficiency of AN2's capital resources and need for additional
capital to achieve its goals; and other risks, including those
described under the heading “Risk Factors” in AN2's reports to be
filed with the SEC, including AN2’s Report on Form 10-Q for the
quarter ended June 30, 2023. These filings, when available, are
available on the investor relations section of our website at
investor.an2therapeutics.com and on the SEC's website at
www.sec.gov. Forward-looking statements contained in this press
release are made as of this date, and AN2 undertakes no duty to
update such information except as required under applicable
law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231010060919/en/
COMPANY CONTACT: Lucy O. Day Chief Financial Officer
l.day@an2therapeutics.com INVESTOR AND MEDIA CONTACT: Anne
Bowdidge ir@an2therapeutics.com
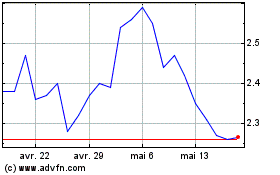
AN2 Therapeutics (NASDAQ:ANTX)
Graphique Historique de l'Action
De Avr 2024 à Mai 2024
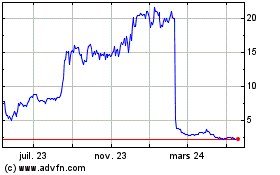
AN2 Therapeutics (NASDAQ:ANTX)
Graphique Historique de l'Action
De Mai 2023 à Mai 2024