Venn Life Sciences Holdings PLC Major contract win (3595H)
16 Mai 2014 - 1:25PM
UK Regulatory
TIDMVENN
RNS Number : 3595H
Venn Life Sciences Holdings PLC
16 May 2014
Venn Life Sciences Holdings Plc
("Venn" or the "Company")
Major contract win
Venn Life Sciences Signs Master Service Agreement for
Multi-Country Study
Value is in excess of EUR3m covering 8 countries in Europe &
North America over 18 months
Venn Life Sciences (AIM: VENN), a growing Clinical Research
Organisation providing clinical trial management solutions and
resourcing solutions to pharmaceutical, biotechnology and medical
device clients, announces the securing of a multi-country
multi-million euro clinical trials management agreement with a
major European biotechnology company.
This multi-site agreement will run for over a year and a half,
covering seven European countries as well as a number of US sites
and is valued in excess of EUR3m. The clinical trial management
programme is for an orphan drug in a rare acute disease area and is
proceeding this month.
Over the past year Venn has increased its geographic footprint
in Europe with new offices in the UK and Germany. Business
development has been focused on key growth areas of drug and device
development and on winning similar studies across Europe, with a
solid pipeline of work for 2014 and 2015 now secured. Studies in
rare diseases have increased significantly in recent years and Venn
has invested in ensuring they have the critical multi-site agile
service capabilities essential to conduct and manage trials
efficiently in this sector.
Commenting, Venn CEO, Tony Richardson, said:
"We are hearing from many of our clients - in the key
pharmaceutical, biotechnology and medical device sectors - of a
growing need for a much more agile multi-country clinical trial
management capability, especially in the still complex greater
European region. Venn has invested heavily in 2013 in preparing the
business for a step change in the size and complexity of contracts
undertaken. This contract win, and our current pipeline, represent
a significant endorsement of our investment strategy since IPO. We
have now moved our business focus from small single country
projects to larger multicentred projects and we expect to cement
this progress with additional project wins in the coming
months."
Enquiries:
Venn Life Sciences Holdings Plc www.vennlifesciences.com
Tony Richardson, Chief Executive Officer Tel: +353 154 99 341
Orla McGuinness, Marketing Manager Tel: +33 (0)1 30 82 67 07
Zeus Capital (Nominated Adviser and
Broker)
Ross Andrews/Andrew Jones (Corporate Tel: 0161 831 1512
Finance)
Dominic Wilson (Institutional Sales) Tel: 020 7533 7727
Walbrook PR Ltd Tel: 020 7933 8787 or venn@walbrookpr.com
Paul McManus Mob: 07980 541 893
Lianne Cawthorne Mob: 07584 391 303
About Venn Life Sciences Limited:
Venn Life Sciences is a Clinical Research Organisation providing
clinical trial management solutions and resourcing solutions to
pharmaceutical, biotechnology and medical device organisations.
With dedicated operations in France, Germany, the Netherlands, the
UK and Ireland and Europe wide representation - Venn specialises in
rapid deployment and management of multisite projects. Venn
recently established an innovation division - innoVenn - focused
primarily on breakthrough development opportunities in Skin
Science.
This information is provided by RNS
The company news service from the London Stock Exchange
END
CNTBBGDUSDBBGSL
Hvivo (LSE:HVO)
Graphique Historique de l'Action
De Juin 2024 à Juil 2024
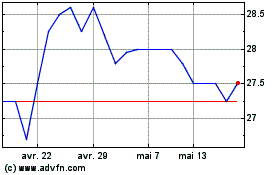
Hvivo (LSE:HVO)
Graphique Historique de l'Action
De Juil 2023 à Juil 2024