ABIONYX Pharma Announces That the French Drug Safety Agency (Agence Nationale de Sécurité du Médicament or ANSM) Has Granted a Compassionate Access Authorization for the bio-HDL (CER-001) in COVID-19 Disease
03 Janvier 2022 - 6:14PM
Business Wire
ABIONYX Pharma (FR0012616852 – ABNX – PEA PME
eligible), a new generation biotech company dedicated to the
discovery and development of innovative therapies for patients,
today announces that the French Drug Safety Agency (Agence
Nationale de Sécurité du Médicament or ANSM), granted a
Compassionate Access Authorization for the bio-HDL (CER-001) in
COVID-19 disease.
Access the complete press release
About ABIONYX Pharma
ABIONYX Pharma is a new generation biotech company that aims to
contribute to health through innovative therapies in indications
where there is no effective or existing treatment, even the rarest
ones. Thanks to its partners in research, medicine,
biopharmaceuticals and shareholding, the company innovates on a
daily basis to propose drugs for the treatment of renal and
ophthalmological diseases, or new HDL vectors used for targeted
drug delivery.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220103005317/en/
NewCap Investor relations Louis-Victor Delouvrier
abionyx@newcap.eu +33 (0)1 44 71 98 53
NewCap Media relations Arthur Rouillé abionyx@newcap.eu
+33 (0)1 44 71 00 15
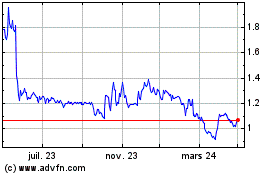
Abionyx Pharma (EU:ABNX)
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
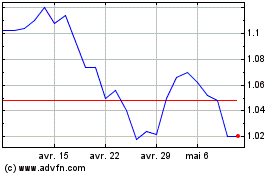
Abionyx Pharma (EU:ABNX)
Graphique Historique de l'Action
De Avr 2023 à Avr 2024