Genomic Vision Technology to Be Tested by NIST (US Gaithersburg) for Establishing Standards Methods for Genome Editing Based ...
21 Janvier 2020 - 7:30AM
Business Wire
Regulatory News:
Genomic Vision (Paris:GV) (FR0011799907 – GV), a
biotechnology company developing molecular assays and Artificial
Intelligence tools to control quality and safety of genetically
modified genomes, today announced that its Molecular Combing
technology will be used by the US National Institute of Standards
and Technology (NIST) to characterize and quantify intended and
unintended genome editing events in NIST’s control material.
The Molecular Combing technology is currently used for
monitoring DNA replication in cancerous cell, for early cancer
detection and the diagnosis of genetic diseases. Genomic Vision has
been part of the NIST Genome Editing Consortium since March 2019.
The consortium’s goal is to address pre-competitive measurement
needs which may eventually help to speed the process of bringing
genome editing products to the market. The Consortium has 33 formal
members and three working groups. Genomic Vision participates in
the first working group, known as “Specificity” which aims to
qualify the genomic assays used for evaluating genome editing
outputs, and to design the control materials needed to increase
confidence in genome editing.
On October 18th, 2019, working group projects were defined and
it was decided that Genomic Vision will use their Quality Control
Assay (QCA) to confirm mid-size to large variants engineered into
cells as controls. Indeed, Genomic Vision FiberVision® platform
provides a powerful quality control tool for the safety and the
optimization of gene editing projects thanks to its high
sensitivity and digital quantification capacity. This platform
allows an unbiased assessment of genetic events through the direct
visualization on single DNA molecules. Combing technology requires
no DNA amplification and completes NGS/PCR based assays.
Samantha Maragh, Leader of the Genome Editing Program at the
NIST, stated: “One of NIST’s missions consists of playing a
pivotal role in helping to define the measurements and standards
for cellular and gene therapies and other regenerative medicine. I
am glad to have innovative industry partners participating in the
Consortium who can apply their technology to solving some of the
measurement challenges of genome editing.”
The National Institute of Standards and Technology (NIST) was
founded in 1901 and is now part of the U.S. Department of Commerce.
NIST is one of the nation’s oldest physical science laboratories,
and addresses measurement research needs in multiple fields
including chemistry, IT, engineering and biology. The NIST Genome
Editing Consortium addresses the measurements and standards needed
to increase confidence and lower the risk of utilizing genome
editing technologies in research and commercial products
(www.nist.gov).
Stéphane Altaba, Chief Operating Officer of Genomic Vision,
commented: “Implementing our technology in the scope of the
NIST consortium into specific work packages will confirm the value
of the molecular combing in detecting and quantifying unwanted
events. The work we have performed in collaboration with Editas
Medicine (Cecilia Cotta-Ramusino et al., CSHL Meeting: Genome
Engineering: The CRISPR-Cas9 Revolution, July 21-23, 2017) and for
other gene editing companies already demonstrated the relevance of
our technology in this field but we wanted to take the time to go
along the regulatory pathway and join the NIST Consortium. We
appreciate the way that this initiative will enable us to interact
on a regular basis with all the major players in gene editing and
will give us the opportunity to use our technology to set up
standard tools. It is exciting to contribute to the fabulous story
of gene editing.”
ABOUT GENOMIC VISION GENOMIC VISION is a biotechnology
company developing molecular assays and Artificial Intelligence
tools to control quality and safety of genetically modified
genomes, in particular in genome editing technologies and
biomanufacturing processes. These tools are currently used for
monitoring DNA replication in cancerous cells, for early cancer
detection, and the diagnosis of genetic diseases. Based near Paris,
in Bagneux, the Company has approximately 30 employees. GENOMIC
VISION is a publicly listed company in compartment C of Euronext’s
regulated market in Paris (Euronext: GV – ISIN: FR0011799907).
For further information, please visit www.genomicvision.com.
Member of CAC® Mid & Small, CAC® All-Tradable and
EnterNext© PEA-PME 150 indexes
FORWARD LOOKING STATEMENT This press release contains
implicitly or explicitly certain forward-looking statements
concerning Genomic Vision and its business. Such forward-looking
statements are based on assumptions that Genomic Vision considers
to be reasonable. However, there can be no assurance that such
forward-looking statements will be verified, which statements are
subject to numerous risks, including the risks set forth in the
“Risk Factors” section of the reference document dated March 28,
2017, available on the web site of Genomic Vision
(www.genomicvision.com) and to the development of economic
conditions, financial markets and the markets in which Genomic
Vision operates. The forward-looking statements contained in this
press release are also subject to risks not yet known to Genomic
Vision or not currently considered material by Genomic Vision. The
occurrence of all or part of such risks could cause actual results,
financial conditions, performance or achievements of Genomic Vision
to be materially different from such forward-looking statements.
This press release and the information contained herein do not
constitute and should not be construed as an offer or an invitation
to sell or subscribe, or the solicitation of any order or
invitation to purchase or subscribe for Genomic Vision shares in
any country. The distribution of this press release in certain
countries may be a breach of applicable laws. The persons in
possession of this press release must inquire about any local
restrictions and comply with these restrictions.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200120005534/en/
Genomic Vision Aaron Bensimon Co-founder, Chairman &
CEO +33 1 49 08 07 50 investisseurs@genomicvision.com
Ulysse Communication Bruno Arabian +33 1 42 68 29 70
barabian@ulysse-communication.com
NewCap Investor Relations / Strategic Communications +33
1 44 71 94 92 gv@newcap.eu
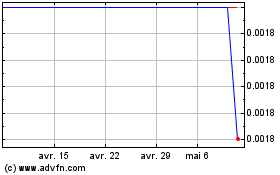
Genomic Vision (EU:GV)
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
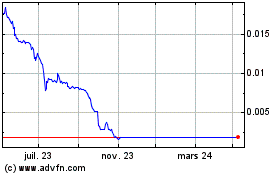
Genomic Vision (EU:GV)
Graphique Historique de l'Action
De Avr 2023 à Avr 2024