Neovacs announces its clinical advisory board meeting to design the Phase III Study for IFNalpha Kinoide in lupus - Paris Feb...
31 Janvier 2019 - 5:45PM

Neovacs announces its clinical advisory board meeting to design the
Phase III Study for IFNalpha Kinoide in lupus - Paris February 11th
PRESS RELEASE ·
PRESS RELEASE · PRESS
RELEASE
NEOVACS ANNOUNCES ITS CLINICAL ADVISORY
BOARD MEETING TO DESIGN THE PHASE III STUDY FOR
IFN-K IN LUPUS
MEETING TO BE HELD IN PARIS,
FEBRUARY 11TH
Paris and Boston, January 31th, 2019 -
5:45 pm CET - Neovacs (Euronext Paris Growth
Paris: ALNEV), leader in active immunotherapy for the
treatment of auto-immune diseases, announces it has formed its
Clinical Advisory Board (CAB) which will meet in Paris on the 11th
of February. Prof Frédéric Houssiau, MD, PhD
(Vice-Rector of the Health Sciences Sector, at the University
Catholic of Louvain, Brussels, Belgium, formerly Head of the
Rheumatology Department at the Cliniques Universitaires Saint-Luc
in Brussels) will chair this CAB composed of multidisciplinary
international medical experts in Lupus:
- Pr Eric Morand, MD, PhD, Head of the School of Clinical
Sciences at Monash Health, Monash University and Head of the Monash
Health Rheumatology Unit.
- Pr Thomas Dörner, MD, Professor of Rheumatology and
Hemostaseology at Charité University Hospitals, Berlin,
- Dr Katherine Thanou, MD, Research Affiliate, Arthritis and
Clinical Immunology Research Program, Oklahoma Medical Research
Foundation, USA
- Dr Michael Tee, MD, MHPED, MBA, Vice Chancellor, Medical Center
Manila, Rheumatology Department, UP Manila College of
Medicine
- Dr Edsel Maurice Salvana, MD, PhD, Director, Institute of
Molecular Biology and Biotechnology, National Institutes of Health,
University of the Philippines Manila, Philippines
This CAB under the Chairmanship of Pr
Frédéric Houssiau will support Neovacs in the design of
the Phase III study for IFN-K in Lupus, based on the results of the
Phase IIb study.
About NeovacsListed on Euronext
Growth since 2010, Neovacs is today a leading biotechnology company
focused on an active immunotherapy technology platform (Kinoids)
with applications in autoimmune and/or inflammatory diseases. On
the basis of the company’s proprietary technology for inducing a
polyclonal immune response (covered by four patent families that
potentially run until 2032) Neovacs is focusing its clinical
development efforts on IFNα-Kinoid, an immunotherapy being
developed for the indication of lupus, dermatomyositis and also in
preclinical trial for Type 1 diabetes. Neovacs is also conducting
preclinical development works on other therapeutic vaccines in the
fields of auto-immune diseases, oncology and allergies. The goal of
the Kinoid approach is to enable patients to have access to safe
treatments with efficacy that is sustained in these life-long
diseases. www.neovacs.fr
Contacts
NEOVACS – Corporate Communication &
Investor RelationsCharlène Masson+33 1 53
10 93 00cmasson@neovacs.com
NEWCAP- Media
Annie-Florence Loyer +33 1 44 71 00 12 / + 33
6 88 20 35 59afloyer@newcap.fr Léa Jacquin +33 1
44 71 20 41 / +33 6 58 14 84 66ljacquin@newcap.fr
ORPHEON FINANCE – Financial
Communication and Investor RelationsJames
Palmer +33 7 60 92 77 74j.palmer@orpheonfinance.com
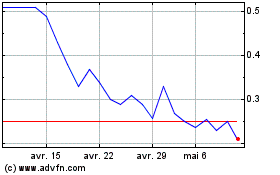
Neovacs (EU:ALNEV)
Graphique Historique de l'Action
De Mar 2024 à Avr 2024
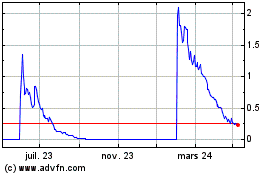
Neovacs (EU:ALNEV)
Graphique Historique de l'Action
De Avr 2023 à Avr 2024
Real-Time news about Neovacs (Euronext): 0 recent articles
Plus d'articles sur