Eurofins Viracor Invests in Innovation with the Launch of Coronavirus (COVID-19) SARS-CoV-2 inSIGHT™ T Cell Immunity Testing
08 Juillet 2021 - 6:42PM
Business Wire
As one of the first commercial
labs to deliver COVID-19 testing, Eurofins Viracor, Inc. continues
to innovate with the launch of Coronavirus SARS-CoV-2 inSIGHT™ T
Cell Immunity testing. Viracor’s inSIGHT™ T Cell Immunity test
delivers an understanding of a patient's response to viral antigens
providing critical insight to aid in treatment decisions. The test
measures CD4+ and CD8+ T Cell response to the SARS-CoV-2 spike (S)
and nucleocapsid (N) proteins to independently evaluate
cell-mediated immunity to Coronavirus SARS-CoV-2 that causes
COVID-19.
Utilizing flow cytometry and
intracellular cytokine staining, SARS-CoV-2 inSIGHT™ testing could
be an important tool for evaluating immunological memory to the
SARS-CoV-2 virus, and even indicate a level of protection from
further infection.
SARS-CoV-2 inSIGHT™ testing
joins a robust menu of COVID-19 testing, including the
recently-launched cPASS™ Coronavirus SARS-CoV-2 Neutralizing
Antibody test. When used in combination with the Neutralizing
Antibody test, results from the tests could help physicians
evaluate two areas of our adaptive immune system that can indicate
immunity. When neutralizing antibody response begins to diminish
months after exposure to the virus or vaccine, the presence of T
cell immunity may signify long-term immunity.
A leader in infectious disease
testing for over 35 years, Viracor has launched molecular and
serological tests to aid in the evaluation of naturally infected or
vaccinated individuals. To see the full list of testing, visit
https://www.eurofins-viracor.com/clinical/our-testing/.
Furthermore, Viracor's SARS-CoV-2 RT-PCR assay offers the best
sensitivity of the 117 laboratories that have submitted results to
FDA's SARS-CoV-2 Reference Panel, with a limit of detection of 180
NAAT Detectable Units/mL1,2.
1 This test has not been cleared or approved
for diagnostic use by the U.S. Food and Drug Administration. This
test has been authorized by FDA under an EUA for use by authorized
laboratories. This test has been authorized only for the detection
of RNA from SARS-CoV-2 virus and diagnosis of SARS-CoV-2 virus
infection, not for any other viruses or pathogens. This test is
only authorized for the duration of the declaration that
circumstances exist justifying the authorization of the emergency
use of in vitro diagnostic tests for detection of SARS-CoV-2 virus
and/or diagnosis of SARS-CoV-2 virus infection under section
564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the
authorization is terminated or revoked sooner.
2 https://www.fda.gov/medical-devices/coronavirus-covid-19-and-medical-devices/sars-cov-2-reference-panel-comparative-data
To learn more, please visit
https://www.eurofins-viracor.com/clinical/news-and-events/categories/news/
For more information, please visit
https://www.eurofins-viracor.com/clinical/
About Viracor Eurofins Viracor has over 30 years of
diagnostic expertise in infectious disease, immunology and allergy
testing for immunocompromised and critical patients. Eurofins
Viracor is a subsidiary of Eurofins Scientific (EUFI.PA), a global
leader in bio-analytical testing, and one of the world leaders in
genomic services. For more information, please visit
https://www.eurofins.com/ and
https://www.viracor-eurofins.com/clinical-diagnostics/.
About Eurofins – the global leader in bio-analysis
Eurofins is Testing for Life. With over 50,000 staff across a
network of more than 800 laboratories in over 50 countries,
Eurofins’ companies offer a portfolio of over 200,000 analytical
methods.
Eurofins Shares are listed on Euronext Paris Stock Exchange.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210708005754/en/
Sally Maysent Director of Marketing Email:
SallyMaysent@Eurofins-Viracor.com
Eurofins Scientific (EU:ERF)
Graphique Historique de l'Action
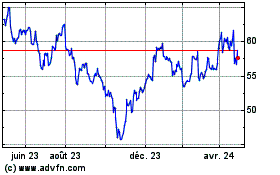
De Mar 2024 à Avr 2024
Eurofins Scientific (EU:ERF)
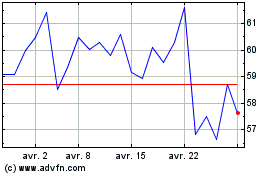
Graphique Historique de l'Action
De Avr 2023 à Avr 2024