Edgewise Gets FDA Orphan, Rare-Pediatric-Disease Designations for Muscular-Dystrophy Program
30 Novembre 2023 - 2:56PM
Dow Jones News
By Colin Kellaher
Edgewise Therapeutics has won a pair of key U.S. Food & Drug
Administration designations for its EDG-5506 muscular-dystrophy
program.
The Boulder, Colo., clinical-stage biopharmaceutical company on
Thursday said the FDA granted orphan-drug designation to EDG-5506
for the treatment of Duchenne and Becker muscular dystrophy, and
rare-pediatric-disease designation for the treatment of
Duchenne.
The FDA's orphan-drug program gives special status to drugs and
biologics for diseases and disorders that affect fewer than 200,000
people in the U.S. and provides for an extended marketing
exclusivity period against competition.
The agency awards priority-review vouchers to companies upon
approval of drugs that are granted the rare-pediatric-disease
designation, and those vouchers can be used to obtain priority
review for another drug or sold to other companies.
Edgewise is currently advancing EDG-5506, a small molecule
designed to prevent contraction-induced muscle damage in
dystrophinopathies including Duchenne and Becker, in multiple Phase
2 studies.
There are currently no cures for Duchenne or Becker muscular
dystrophy, which are neuromuscular genetic diseases.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
November 30, 2023 08:41 ET (13:41 GMT)
Copyright (c) 2023 Dow Jones & Company, Inc.
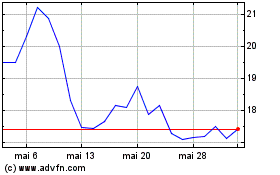
Edgewise Therapeutics (NASDAQ:EWTX)
Graphique Historique de l'Action
De Août 2024 à Sept 2024
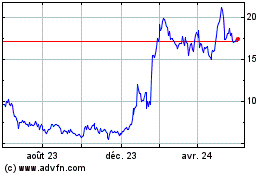
Edgewise Therapeutics (NASDAQ:EWTX)
Graphique Historique de l'Action
De Sept 2023 à Sept 2024