Delcath Systems to Host Fourth Quarter and Full Year 2024 Earnings Call
20 Février 2025 - 2:30PM
Business Wire
Delcath Systems, Inc. (Nasdaq: DCTH), an interventional oncology
company focused on the treatment of primary and metastatic cancers
of the liver, announced today it will host a conference call on
March 6, 2025, at 8:30 AM Eastern Time to discuss results for its
fourth quarter and full year ended December 31, 2024.
Conference Call Information
To participate in this event, dial in approximately 5 to 10
minutes before the beginning of the call.
Event Date: Thursday, March 6, 2025 Time: 8:30 AM Eastern
Time
Participant Numbers: Toll Free: 1-877-407-3982 International:
1-201-493-6780 Webcast:
https://viavid.webcasts.com/starthere.jsp?ei=1706497&tp_key=9763647546
A replay of the webinar will be available shortly after the
conclusion of the call and will be archived on the company's
website here.
About Delcath Systems, Inc., HEPZATO KIT and CHEMOSAT
Delcath Systems, Inc. is an interventional oncology company
focused on the treatment of primary and metastatic liver cancers.
The company's proprietary products, HEPZATO KIT™ (Hepzato
(melphalan) for Injection/Hepatic Delivery System) and CHEMOSAT®
Hepatic Delivery System for Melphalan percutaneous hepatic
perfusion (PHP), are designed to administer high-dose chemotherapy
to the liver while controlling systemic exposure and associated
side effects during a PHP procedure.
In the United States, HEPZATO KIT is considered a combination
drug and device product and is regulated and approved for sale as a
drug by the FDA. HEPZATO KIT is comprised of the chemotherapeutic
drug melphalan and Delcath's proprietary Hepatic Delivery System
(HDS). The HDS is used to surgically isolate the liver while
simultaneously filtrating hepatic venous blood during melphalan
infusion and washout. The use of the HDS results in loco-regional
delivery of a relatively high melphalan dose, which can potentially
induce a clinically meaningful tumor response with minimal
hepatotoxicity and reduce systemic exposure. HEPZATO KIT is
approved in the United States as a liver-directed treatment for
adult patients with metastatic uveal melanoma (mUM) with
unresectable hepatic metastases affecting less than 50% of the
liver and no extrahepatic disease, or extrahepatic disease limited
to the bone, lymph nodes, subcutaneous tissues, or lung that is
amenable to resection or radiation. Please see the full Prescribing
Information, including BOXED WARNING for the HEPZATO KIT.
In Europe, the device-only configuration of the HDS is regulated
as a Class III medical device and is approved for sale under the
trade name CHEMOSAT Hepatic Delivery System for Melphalan, or
CHEMOSAT, where it has been used in the conduct of percutaneous
hepatic perfusion procedures at major medical centers to treat a
wide range of cancers of the liver.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20250220212395/en/
Investor Relations Contact: ICR Healthcare
investorrelations@delcath.com
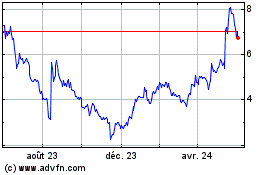
Delcath Systems (NASDAQ:DCTH)
Graphique Historique de l'Action
De Jan 2025 à Fév 2025
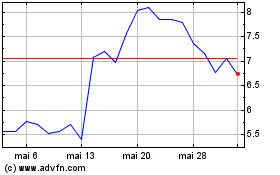
Delcath Systems (NASDAQ:DCTH)
Graphique Historique de l'Action
De Fév 2024 à Fév 2025